Choreographing the microRNA-target dance

DALLAS – January 19, 2017 – Scientists face a conundrum in their quest to understand how microRNAs regulate genes and therefore how they influence human disease at the molecular level: How do these tiny RNA molecules find their partners, called messenger RNAs, on a crowded cellular dancefloor?
MicroRNAs pair up with messenger RNAs to turn down the production of proteins. But there are far more of the messenger RNAs in the crowd, leaving scientists to ponder how the microRNAs can efficiently regulate a large excess of messenger RNA partners.
Molecular biologists at UT Southwestern Medical Center were able to uncover a new mechanism that choreographs this complex molecular dance by applying the latest in gene editing technology combined with a traditional method of making a microRNA target produce a fluorescent green protein. The successful “dance move” – called Argonaute phosphorylation – enables a microRNA to switch messenger RNA dance partners more efficiently.
“Our research addresses a fundamental question regarding how a microRNA is able to regulate a large set of target messenger RNAs, even though the microRNA is greatly outnumbered,” said Dr. Joshua Mendell, Professor of Molecular Biology and a Howard Hughes Medical Institute Investigator at UT Southwestern.
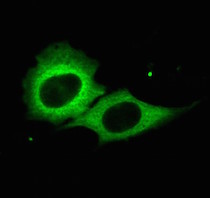
The microRNA pathway is critically important to health and disease, serving as a kind of volume control for genes, dialing down the expression of specific proteins, said Dr. Mendell, a CPRIT Scholar in Cancer Research. UTSW researchers have previously found, for example, that defects in the microRNA pathway contribute to certain childhood cancers and specific microRNAs can accelerate or inhibit cancer by regulating tumor suppressor or tumor promoting genes. MicroRNAs play important roles in many other diseases including heart disease.
CRISPR gene editing technology allowed the scientists to switch off a different gene in each cell across millions of cells. Cells became more fluorescent when genes that impacted the microRNA pathway were switched off, leading scientists to the discovery of the new phosphorylation mechanism involved in controlling microRNA-target interactions.
“This research uncovered a new and fundamental aspect of the microRNA pathway in which phosphate molecules are rapidly added and removed from key proteins in the pathway. We believe this mechanism allows microRNAs to engage target messenger RNAs, silence them, and then efficiently move on to the next target,” said first author Ryan Golden, a student in the Medical Scientist Training Program at UT Southwestern and a member of the Mendell lab.
Find out more
|
In addition to shedding new light on the microRNA pathway, researchers say the distinctive combination of techniques used to decipher the pathway should be widely applicable to other biological questions, allowing labs to quickly identify critical components of important genetic pathways.
“This study represents the first time this experimental strategy has been used to study the microRNA pathway on a genome-wide scale. It is a very powerful approach. This work lays out a methodology that could be used to study many different biomedical problems,” said Dr. Mendell, a member of the Harold C. Simmons Comprehensive Cancer Center.
The work, published in the journal Nature, was supported by the Howard Hughes Medical Institute and grants from Cancer Prevention and Research Institute of Texas (CPRIT), the National Institutes of Health, the Cancer Research Institute, and the Leopoldina Fellowship Program from the German National Academy of Sciences Leopoldina.
Collaborators included researchers with the Eugene McDermott Center for Human Growth and Development; Next Generation Sequencing Core; the UT Southwestern Flow Cytometry Core; the UT Southwestern Protein Chemistry Technology Core; the Quantitative Biomedical Research Center; the Harold C. Simmons Comprehensive Cancer Center; the Hamon Center for Regenerative Science and Medicine; and the Center for the Genetics of Host Defense.
Researchers were Ryan J. Golden, first author and graduate student researcher in the Medical Scientist Training Program; graduate student researchers Hema Manjunath, Andres Ramirez-Martinez, and Jiaxi Wu; postdoctoral researchers Dr. Tuo Li, Dr. Juliane Braun, and Dr. Florian Kopp; Beibei Chen, computational biologist; Dr. Xiang Chen, research specialist; Vanessa Schmid, manager of the McDermott Center Next Generation Sequencing Core; Dr. Tsung-Cheng Chang, Assistant Professor of Molecular Biology; Dr. Vincent Tagliabracci, Assistant Professor of Molecular Biology and Michael L. Rosenberg Scholar in Medical Research; Dr. Yang Xie, Associate Professor of Clinical Sciences; Dr. Zhijian Chen, Professor of Molecular Biology and the Center for the Genetics of Host Defense, Howard Hughes Medical Institute Investigator, who holds the George L. MacGregor Distinguished Chair in Biomedical Science.
Summary Q&A: Argonaute phosphorylation cycleBy Dr. Ryan Golden with the Medical Scientist Training Program and Dr. Joshua Mendell in the Department of Molecular Biology at UT Southwestern What did you find?Our laboratory has a long-standing interest in the microRNA pathway, a fundamental mechanism through which cells control gene expression. Proper activity of the microRNA pathway is critical for normal development and physiology and abnormal microRNA function contributes to many human diseases including cancer and cardiovascular disease. To better understand how microRNAs function and how the pathway is regulated, we applied a new method based on CRISPR-mediated genome editing that allowed us to simultaneously test virtually all human genes for a role in the microRNA pathway. This unexpectedly revealed that the central mediator of microRNA activity, a protein called Argonaute, requires the constant addition and removal of phosphate molecules on specific amino acids in order for microRNAs to efficiently regulate gene expression. We believe that this phosphorylation cycle allows Argonaute to efficiently interact with and regulate a target messenger RNA, and then quickly move on to a new target. Why is that important?Given the critical roles of the microRNA pathway in normal physiology, it is important to fully understand how the pathway works at the molecular level since defects in microRNA function can contribute to diverse disease states. Our improved understanding of how microRNAs work could one day allow manipulation of the pathway for therapeutic benefit. In addition, this work represents an important technical advance as it provides a roadmap for using cutting-edge CRISPR methods to dissect the mechanisms of pathways that control gene expression in human cells. What diseases does the research apply to and how would the findings eventually help in preventing those diseases?Our laboratory and many others have demonstrated that abnormalities in the microRNA pathway cause diverse human diseases including cancer. By identifying a previously unidentified mechanism that acts as a major regulator of microRNA activity, our findings reveal a new aspect of the pathway that might be used by cells to control microRNA function both in normal physiology as well as in disease. An important next step in our research will be to examine this newly defined Argonaute phosphorylation cycle in normal and diseased tissues to determine whether it is activated or inactivated in these settings. How does this help other researchers in the field?The Argonaute phosphorylation cycle that we identified reveals a critical control mechanism for the microRNA pathway, but also raises many important questions for the field. For example, are there circumstances in which normal or disease cells engage this pathway to regulate microRNA activity? Can we take advantage of this mechanism to activate or inhibit microRNA activity for therapeutic benefit in some contexts? Do other gene expression regulatory pathways use similar phosphorylation events to regulate their activity? |
About UT Southwestern Medical Center
UT Southwestern, one of the premier academic medical centers in the nation, integrates pioneering biomedical research with exceptional clinical care and education. The institution’s faculty includes many distinguished members, including six who have been awarded Nobel Prizes since 1985. The faculty of almost 2,800 is responsible for groundbreaking medical advances and is committed to translating science-driven research quickly to new clinical treatments. UT Southwestern physicians provide medical care in about 80 specialties to more than 100,000 hospitalized patients and oversee approximately 2.2 million outpatient visits a year.
###
Media contact: Russell Rian
214-648-3404
russell.rian@utsouthwestern.edu
To automatically receive news releases from UT Southwestern via email, subscribe at www.utsouthwestern.edu/receivenews