Center for Alzheimer’s and Neurodegenerative Diseases
The Center for Alzheimer's and Neurodegenerative Diseases (CAND) was established in 2014 to develop mechanism-based approaches to diagnose and treat Alzheimer's disease and related disorders.
Founded and directed by Marc Diamond, M.D., a neurologist with over 15 years of experience caring for patients with neurodegenerative diseases, the center focuses on the basic mechanisms underlying these conditions, especially tauopathies. These disorders are caused by the accumulation of tau protein into ordered assemblies (amyloids). Other self-assembling proteins such as b-amyloid-beta, a-synuclein, and TDP-43, responsible for diseases like Parkinson's and amyotrophic lateral sclerosis, are also a major research focus at CAND.
Neurodegenerative diseases involve molecular abnormalities that progressively impair brain function. To fully understand these processes, research must span biological scale, from protein structure to pathophysiology in patients. The most impactful discoveries have come from combining disciplines, such as linking biochemical events with in vivo outcomes. Consequently the CAND brings together a diverse, collaborative group of scientists, including 9 primary and 5 affiliated faculty members, approximately 60 students/postdocs, 20 scientific staff, and full administrative support, all within a 25,000 square-foot space equipped with dedicated biophysics, cell culture, and animal research facilities. CAND researchers also work closely with clinician-investigators at the Cognitive and Behavioral Clinic at UT Southwestern and beyond, using patient samples to test new ideas. The goal is to develop diagnostics and therapies to detect and treat disease before it causes brain dysfunction.
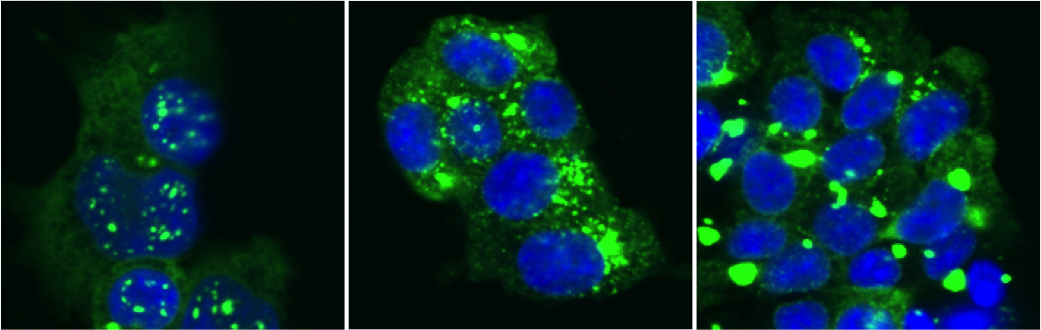
Many proteins associated with adult-onset neurodegenerative diseases share similar self-assembly properties, forming highly ordered amyloid fibrils rich in cross-beta sheet structure. The Diamond lab was the first to propose that prion mechanisms drive the diversity and progression of tau-related disorders. In this model, tau assemblies of defined structure spread from cell to cell, serving as a templates for further aggregation. The diversity of tau assembly structures underlies distinct pathological and clinical presentations of disease. Multidisciplinary efforts have been required to study the initial protein folding events that lead to pathology, and the structural, biochemical, and cellular basis of disease progression. These insights have guided new strategies to target pathological amyloid proteins. Additionally, immune signaling that impacts protein aggregation and propagation is now increasingly recognized to play a role in disease, and is studied within the center, and in the affiliated Neuroinflammation and Neurodegeneration Clinic.
The recognition that common disorders like Alzheimer's, Parkinson's, and amyotrophic lateral sclerosis share key features of aggregate replication and propagation has transformed the study of neurodegenerative diseases. This concept has shaped CAND's research focus and attracted talented students, postdocs, and faculty to an innovative and collaborative environment. Understanding becomes healing. Join us!