UT Southwestern investigators identify missing link in kidney cancer prognosis
DALLAS – June 8, 2018 – Normal cells located next to kidney cancer cells provide new clues about prognosis and survival rates, researchers from UT Southwestern Medical Center’s Kidney Cancer Program report.
Kidney cancer is notorious for its variability. Rampant in some patients, the cancer causes death within months. In others, metastatic kidney cancer can smolder for years without treatment.
Several factors have been identified that help determine how aggressive the cancer is in a particular patient. These factors include blood parameters – red blood cell count, platelet and neutrophil numbers, and calcium levels. In addition, the functional status of the patient and how quickly metastases develop also provide clues to the disease.
However, how the cancer affects the body to induce these changes is poorly understood.
Research published today in Cancer Discovery shed some light. The research was authored by Dr. Tao Wang, Assistant Professor of the Quantitative Biomedical Research Center, Clinical Sciences and in the Center for the Genetics of Host Defense, as well as Bioinformatics Co-Leader of the Kidney Cancer Program, and Dr. James Brugarolas, Sherry Wigley Crow Endowed Chair in Cancer Research, and Director of the Kidney Cancer Program at UT Southwestern Medical Center’s Harold C. Simmons Comprehensive Cancer Center.
Drs. Wang and Brugarolas reasoned that some clues could be found in the tumor neighborhood, the cells surrounding and infiltrating the tumor. However, studying neighboring cells is not easy, as they are intermingled with tumor cells.
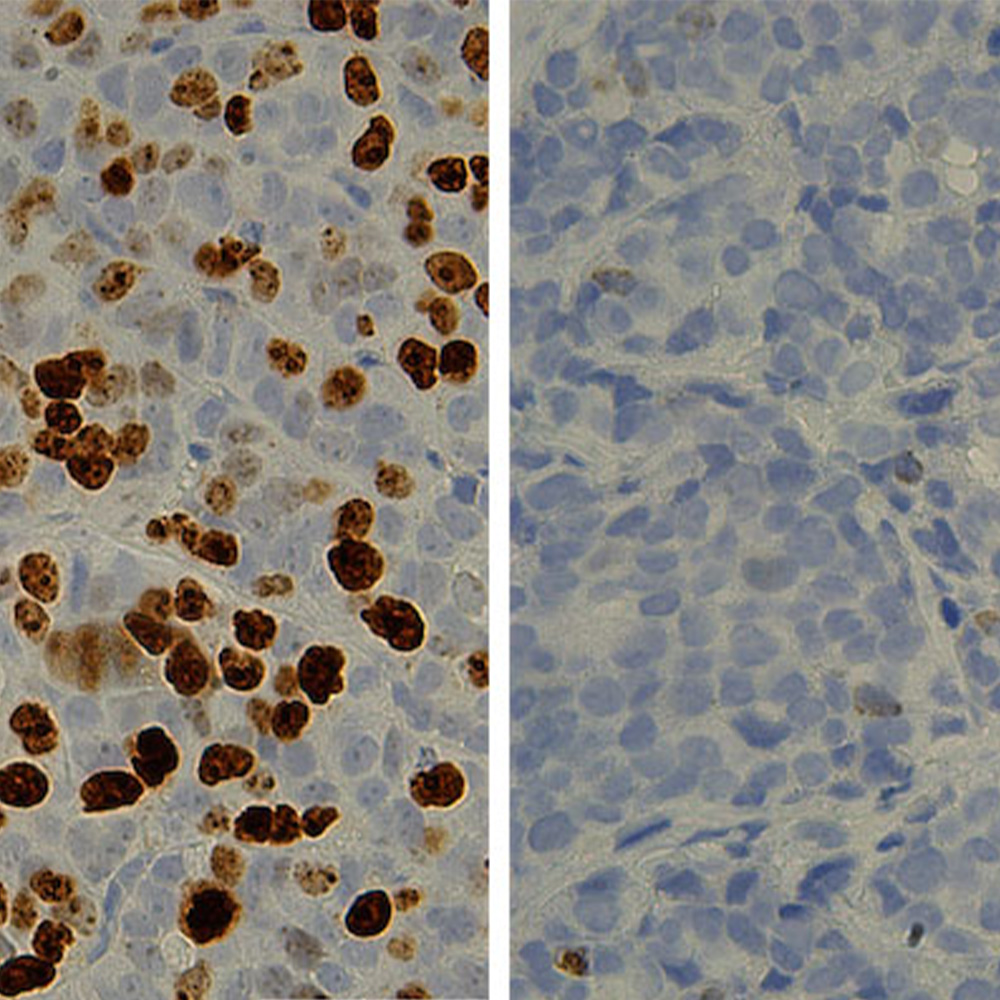
To evaluate the tumor neighborhood, also called the tumor microenvironment (TME), the investigators took an innovative approach. Over the years, Dr. Brugarolas and his team have transplanted kidney cancer samples from hundreds of patients into mice. Some tumors, the most aggressive, will grow in the mouse kidneys, allowing scientists to study them. An important aspect of these tumors is that only the cancer cells expand, while the neighborhood cells are lost. Drs. Brugarolas and Wang reasoned that by comparing patients’ tumors with corresponding tumors growing in mice, they could home in on the neighborhood cells and learn more about them.
The approach worked. Dr. Wang, a bioinformatics and statistics expert, developed a program called DisHet to computationally subtract the tumor growing in the mouse from the patient’s tumor. The difference corresponded to the neighborhood cells, allowing the scientists to identify genes that were turned on in those cells. This approach provided a neighborhood gene signature, or as they called it, an empirically defined tumor microenvironment (eTME).
Dr. Wang and colleagues found twice as many genes in the eTME than previous approaches had discovered. They also determined that over 60 percent of previously assigned neighborhood genes were not abundantly expressed in the microenvironment.
Using this approach, the researchers determined that kidney tumors established one of two different neighborhoods: One neighborhood is characterized by activation of inflammatory genes, and the other is not. They found that tumors with an inflamed neighborhood were more likely to be associated with anemia (low blood cell count) and elevated platelet counts, factors linked to more aggressive cancers. Importantly, the researchers also discovered that those tumors that induced the inflamed neighborhood were associated with worse survival in patients.
However, “tumors with inflamed neighborhoods may respond better to immunotherapy treatment,” noted Dr. Wang.
These new insights into tumor neighborhoods will help doctors better understand their patients’ tumors and may eventually lead to new therapeutic interventions, as well as more informed treatment decisions involving cancer immunotherapy.
“This is a great example of how the Kidney Cancer Program promotes innovative inter-disciplinary research and fosters the careers of young researchers,” said Dr. Yang Xie, Director of the Quantitative Biomedical Research Center and Associate Professor of Clinical Sciences and Bioinformatics.
The project was supported in part by a Specialized Program of Research Excellence (SPORE) award from the National Cancer Institute, one of two such awards for kidney cancer in the country.
Simmons Cancer Center, the only National Cancer Institute-designated Comprehensive Cancer Center in North Texas and one of just 49 NCI-designated Comprehensive Cancer Centers in the nation, includes 13 major cancer care programs. In addition, the Center’s education and training programs support and develop the next generation of cancer researchers and clinicians. The Simmons Cancer Center is among only 30 U.S. cancer research centers to be designated by the NCI as a National Clinical Trials Network Lead Academic Participating Site.
About UT Southwestern Medical Center
UT Southwestern, one of the premier academic medical centers in the nation, integrates pioneering biomedical research with exceptional clinical care and education. The institution’s faculty has received six Nobel Prizes, and includes 22 members of the National Academy of Sciences, 16 members of the National Academy of Medicine, and 15 Howard Hughes Medical Institute Investigators. The faculty of more than 2,700 is responsible for groundbreaking medical advances and is committed to translating science-driven research quickly to new clinical treatments. UT Southwestern physicians provide care in about 80 specialties to more than 100,000 hospitalized patients, 600,000 emergency room cases, and oversee approximately 2.2 million outpatient visits a year.