Fish oil adds secret touch to boost nanoparticle therapy
An experimental nanoparticle therapy that combines low-density lipoproteins (LDL) and fish oil preferentially kills primary liver cancer cells without harming healthy cells, UT Southwestern Medical Center researchers have found.
“This approach offers a potentially new and safe way of treating liver cancer, and possibly other cancers,” said Dr. Ian Corbin, Assistant Professor in the Advanced Imaging Research Center and of Internal Medicine at UT Southwestern. “The method utilizes the cholesterol carrier LDL combined with fish oil to produce a unique nanoparticle that is selectively toxic to cancer cells.”

Primary liver cancer, or hepatocellular carcinoma, is the sixth most prevalent type of cancer and the third-leading cause of cancer-related deaths worldwide, according to the National Cancer Institute.
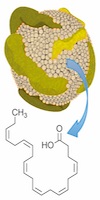
Fish oils are particularly rich in omega-3 fatty acids such as docosahexaenoic acid, also known as DHA. Although several studies have reported an association between cancer prevention and omega-3 fatty acids, the link is not as clear-cut in relation to established tumors, Dr. Corbin said.
In this study, conducted in rats, the newly formulated LDL-DHA nanoparticles were injected into the artery leading to the liver, the site of the cancer, he said.
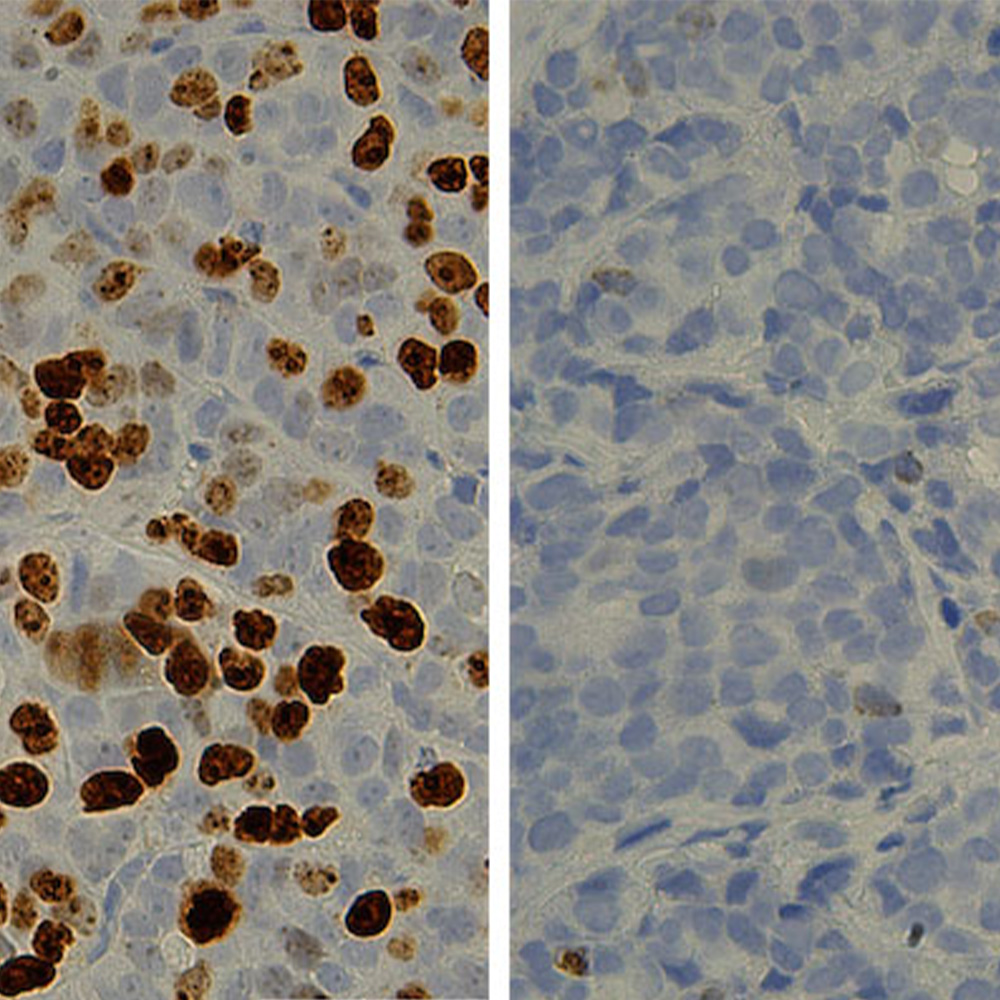
“This research study clearly demonstrates the anticancer potential of omega-3 fatty acids,” he said, adding that while the study showed significant cancer cell toxicity, it is too soon to tell whether the approach is able to kill every cancer cell. Future experiments will examine that question, as well as whether the LDL-DHA strategy improves long-term cancer survival, he added.
Dr. Corbin, a member of the Harold C. Simmons Comprehensive Cancer Center, led a multidisciplinary team spanning the fields of lipid biochemistry, cancer biology, nutritional science, biotechnology, and advanced imaging to create and characterize the novel nanoparticle formulation.