Cryo-electron microscope center zooms in on life at atomic scale
In May, UT Southwestern Medical Center opened a new $17 million cryo-electron microscope (cryo-EM) facility housing a unique collection of instruments that researchers can use to view 3-D images of objects as tiny as an atom all the way up to intact cells.
How the instruments work
Team members in the cryo-electron microscope facility view 3-D images of objects obtained from the high-powered Titan Krios microscope (background).
The Scios DualBeam is a molecular sandblaster that precisely carves very thin slices of cells that can be transferred to and imaged in three dimensions on the Titan Krios. A robotic arm inside the Titan Krios transfers frozen samples from their storage containers into the microscope for viewing while maintaining a vacuum and cryogenic temperatures. The microscope, which is about 12. feet tall and weighs 2 tons, can hold and precisely move 12 samples in an automated manner so that thousands of images can be recorded, processed via computers, and interpreted to generate 3-D images for study. The Talos Arctica is a fully automated but less powerful instrument that enables researchers to screen for optimal conditions before moving on to the Titan Krios. This imaging is facilitated by new high-tech cameras capable of directly recording electrons, unlike conventional cameras that must first convert the electrons’ energy to light, losing sensitivity in the process. |
“We are the only institution in the world with this configuration of instruments,” said Dr. Sandra Schmid, Chair of the Department of Cell Biology and holder of the Cecil H. Green Distinguished Chair in Cellular and Molecular Biology. “It establishes UT Southwestern as one of the world’s top facilities for cryo-EM structural biology.”
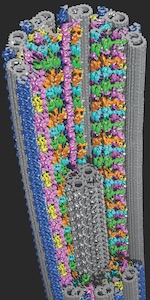
The facility’s three cutting-edge instruments – a Titan Krios, a Talos Arctica, and a Scios DualBeam for thin-slice cryo-electron tomography – provide the technologies to help accelerate UT Southwestern’s biomedical investigations on everything from cancer biology to drug discovery and run 24 hours a day, seven days a week. The instruments represent a significant advancement in the scientific field known as structural biology, and for work at UT Southwestern. By studying 3-D structures at atomic resolution, scientists can uncover new clues about the molecular machinery of cells and how molecules involved in diseases might be targeted with drugs.
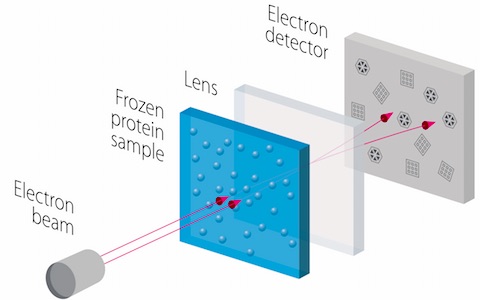
These instruments analyze specimens that have been rapidly frozen to prevent the formation of damaging ice crystals. The specimens are then viewed in special holders under conditions that keep them at cryogenic temperatures (minus 321 degrees Fahrenheit).
The three advanced instruments and the building to house them received funding from an anonymous donor, UT Southwestern, the Cancer Prevention and Research Institute of Texas (CPRIT), the UT System’s Science and Technology Acquisition and Retention program, and the Howard Hughes Medical Institute (HHMI). Because minute temperature changes and sound vibrations from talking can disrupt the highly sensitive instruments, the microscopes are operated remotely and kept in a special climate- and humidity-controlled, vibration-protected building.

“This facility marks a milestone in the evolution of our structural biology research efforts at UT Southwestern. We are grateful to our supporters whose visionary generosity has helped us create this exceptional facility aimed at making fundamental basic discoveries that can be foundational for advances in medicine,” said Dr. Daniel K. Podolsky, President of UT Southwestern, who holds the Philip O’Bryan Montgomery, Jr., M.D. Distinguished Presidential Chair in Academic Administration, and the Doris and Bryan Wildenthal Distinguished Chair in Medical Science.
Cryo-EM and EM tomography allow biological specimens to be viewed in a more lifelike state and environment than X-ray crystallography, the longtime gold standard of structural biology that requires assembling molecules into crystals. Many of biology’s most intriguing molecules have proved notoriously resistant to crystallization, Dr. Schmid explained. Cryo-EM tomography is a more powerful cellular imaging technique to view and study how proteins interact, work, and are spatially arranged within cells – a technology that enhances our understanding of fundamental processes underlying cellular functions integral to the health of all living things.
The new facility is a joint effort of the Departments of Cell Biology and Biophysics. Dr. Schmid; Dr. Michael Rosen, Chair of Biophysics and an HHMI Investigator at UT Southwestern; and Dr. Daniela Nicastro, Associate Professor of Cell Biology and Biophysics and a CPRIT Scholar, spent four years planning and building the facility, which is a shared resource across the academic medical center. Scientists can schedule time on the microscopes and even view their images from remote locations via computer, enabling international collaborations.

“In addition to the unique configuration, we are using these instruments collaboratively to further research campuswide – from basic research to answer fundamental scientific questions to translational investigations in which those answers may result in potential treatments for human disease,” said Dr. Rosen, who has additional appointments in Biochemistry and in the Cecil H. and Ida Green Comprehensive Center for Molecular, Computational, and Systems Biology, and who holds the Mar Nell and F. Andrew Bell Distinguished Chair in Biochemistry.
With funding from a CPRIT grant, Dr. Nicastro is studying mistakes in DNA repair that are thought to drive the development of cancer. “Before you can understand how a process goes wrong, you have to understand how it works,” she explained. “And seeing is believing!”
Cryo-electron Microscopy The technology involves imaging specimens at cryogenic temperatures by electron microscopy. Benefits over X-ray crystallography include the ability to quickly create high-resolution models of molecules sometimes resistant to other approaches. The first reported use of this technology (published in 1975) related to the structure of bacteriorhodopsin. A key advance in the field in the 1980s and 1990s involved the use of liquid ethane to flash-freeze proteins in solution and hold them still. By 2012, electron microscopes evolved that could capture images of a molecule at dozens of frames per second. |