Clinical Trials
As a crucial part of the clinical research process to develop new treatments, improve diagnosis, and help prevent diseases, UT Southwestern conducts clinical studies and clinical trials.
Clinical trials are supported by the Office of Clinical Trial Management (OCTM), the Clinical Research Unit (CRU) and its Investigational Drug Service (IDS), and the Clinical and Translational Science Award (CTSA) Program. Clinical research is also empowered by the vast and diverse research opportunities provided by Dallas County hospital Parkland Health.
Office of Clinical Trials Management

The Office of Clinical Trials Management (OCTM) ensures the sites at which clinical research is performed comply with protocol and regulatory requirements and have the technical, operational, equipment, infrastructure, and staff required for the safe and compliant conduct of the clinical trial.
Activities and services of the OCTM include:
- Overseeing the UTSW Performance Site Approval Process
- Ensuring the appropriate level of research credentialing for study personnel
- Inventorying resources required for submitted protocols
- Determining protocol feasibility for subject population and facility resources
- Coordinating the review of resource utilization across departments/service lines/sites
- Facilitating Study Implementation and Conduct
- Creating research study calendars and order sets
- Assisting the Billing Office in correctly invoicing study activities
- Facilitating communication and education across stakeholders involved in the conduct of the study
- Reviewing serious adverse events (SAE) and unanticipated problems involving risks to subjects or others (UPIRSO) and creating remediation/prevention plans
- Observing study phases, including study closeout
- Supporting Study Teams
- Analyzing project-specific institutional resources
- Reviewing feasibility of proposed interventions and evaluations
- Providing guidance during the approval processes
- Reviewing study budget
Need assistance? OCTM@UTSouthwestern.edu
CTSA Trial Services

The Clinical and Translational Science Award (CTSA) Program website at UT Southwestern provides information on a variety of clinical research services to investigators at each stage of their research including:
Aston Clinical Research Facilities

Aston Facilities are institutional resources, located on U7 and U9 floors of Aston Ambulatory Clinic Building on South Campus, supported and managed by Office of Clinical Research (OCR), which provide exam rooms, infusion chairs, DEXA scans, sample processing and storage facilities, procedure rooms, specialized research equipment rooms, and trained clinical research personnel to handle all aspects of a study.
Clinical Research Unit

The Clinical Research Unit (CRU) provides a controlled environment with trained research staff to support a broad range of research trials. All stages of investigators, from early to experienced, can access CRU services for patient-facing activities including examinations, consultations, infusions, and physiologic testing and can complement or expand the capacity and capabilities of their research teams with nurses, coordinators, pharmacists, and dietitians.
Clinical Research Units include:
Investigational Drug Service

The Investigational Drug Service (IDS) [within Pharmacy Services] provides full research pharmacy services to support clinical trials, including:
- Consulting services to develop pharmacy-related aspects of a protocol
- Secured location for drug storage and dispensing
- Interactive Voice Response System (IVRS)
- Subject randomization and blinding
- Protocol adherence, such as drug counseling and drug accountability
- Initial receipt and inventory of investigational agents
- Preparation and maintenance of drug accountability records
- Meetings with investigators and study monitors
- Preparation of protocol and drug information for pharmacy and study personnel
- Inventory control and accountability audits
- Ordering and maintaining drug stock
- Monitoring visits
- Storage and maintenance of returned drugs
- Reconciliation of drug accountability records, drug return, and maintenance of discontinued study pharmacy products upon study termination
The IDS is staffed by full-time research pharmacists and pharmacy technicians who are available to directly assist investigators. The IDS unit features a clean room, or a low-pollutant environment, for the storage and preparation of medication for clinical trials, where air quality is constantly controlled. An anteroom annex is connected to the clean room. The IDS also stores a limited number of study binders and is equipped to facilitate study monitor visits with advance notice.
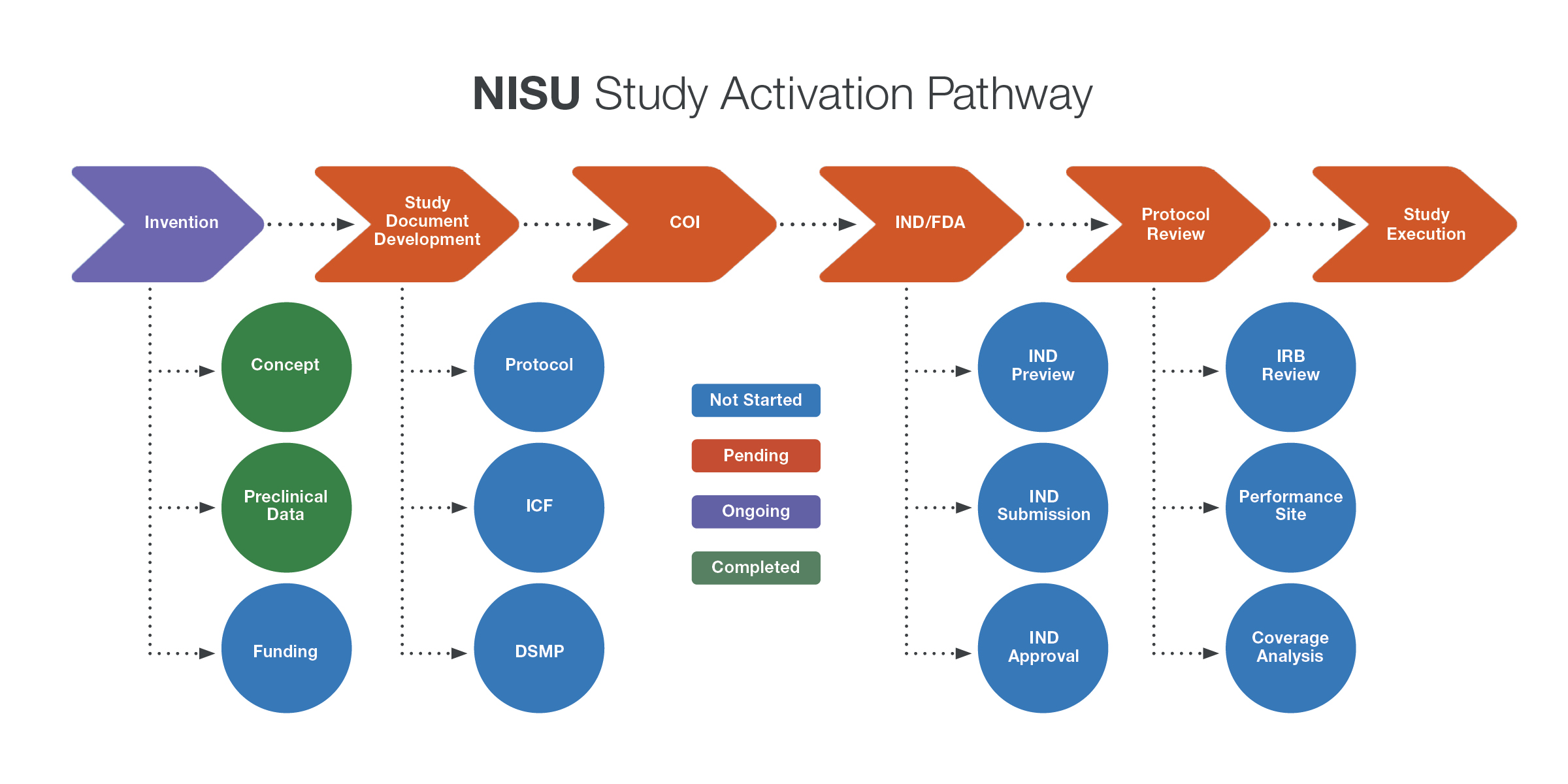
Novel Intervention Support Unit (NISU)

Advancing new therapeutic and diagnostic innovations – such as novel drugs, medical devices, and diagnostic tools – from the lab to the clinic is a complex and resource-intensive process. The Novel Intervention Support Unit (NISU) accelerates this translation by providing comprehensive support to investigators. NISU helps reduce delays in clinical testing, maximize commercialization opportunities, and maintain momentum in translational research.
NISU Services
- Develop personalized activation plans or road maps for investigators preparing to launch first-in-human studies.
- Provide project management and coordination, from invention disclosure to clinical trial activation.
- Collaborate with key institutional offices to bring the invention/discovery into the clinic, either internally or through a commercialization partner.
- Connect with regulatory support, including protocol development, data and safety monitoring plans, Data and Safety Monitoring Board formation, and multisite trial coordination.
Need assistance? Contact Saumya Abraham, Clinical Research Manager, Office of Clinical Research: Saumya.Abraham@UTSouthwestern.edu
Recruitment

Recruitment of clinical research volunteers can be one of the most challenging and time-consuming parts of a research study. The Research Recruitment Office works with study teams to develop and implement innovative recruitment strategies to recruit research volunteers.
Our Services
- Electronic health record-based feasibility assessments and cohort identification
- Recruitment consultations available at project startup, after funding, or when additional support is needed to improve enrollment
- Recruitment Dashboard that enables study teams to sort and filter potentially eligible participants and track recruitment outcomes
- Training on recruitment informatics tools (including i2b2, TriNetX, and SlicerDicer)
- Patient portal recruitment (Epic MyChart)
Need assistance? ResearchRegistry@utsouthwestern.edu
Questions about Clinical Trials?
Office of Clinical Research