Type of KRAS mutation may guide more effective cancer treatments
UTSW-led study shows that different mutations of the gene affect how tumors interact with the immune system
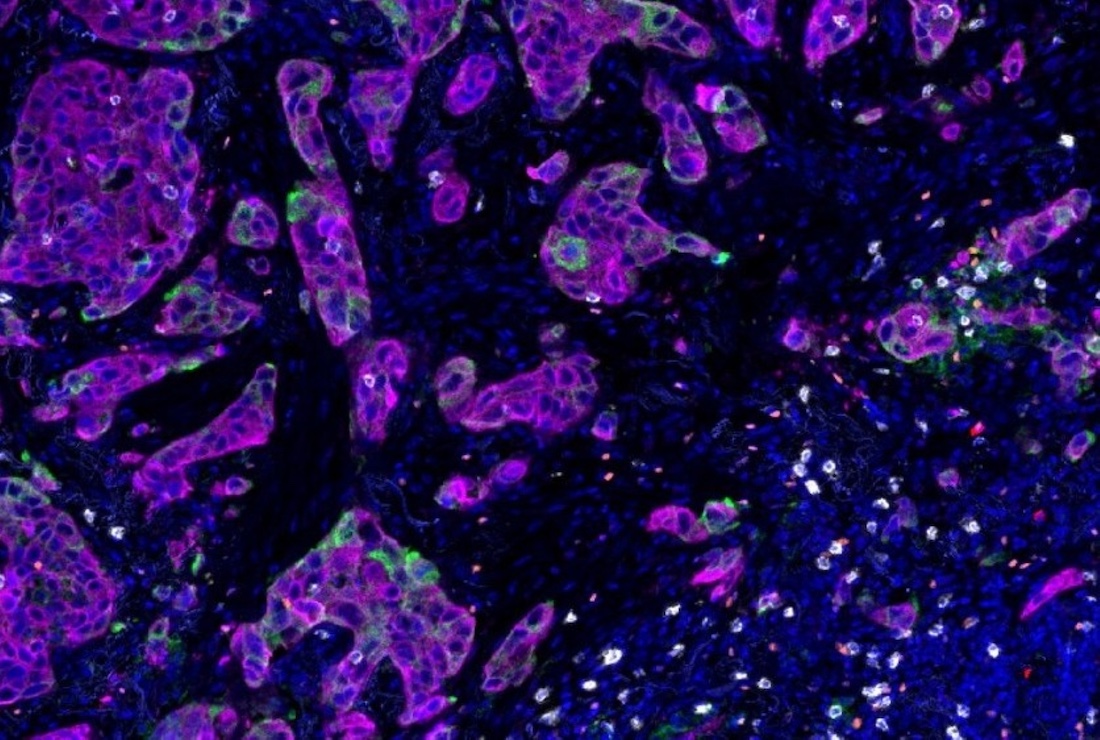
DALLAS – Feb. 18, 2026 – KRAS is the most frequently mutated oncogene across all human cancers. Although different KRAS mutations have long been thought to exert the same cancer-driving effects, a new study led by UT Southwestern Medical Center researchers suggests that different KRAS mutation types can variously impact how cancer cells interact with immune cells, significantly affecting the malignant cells’ behavior. The findings, published in Science Translational Medicine, could lead to personalized therapies based on the KRAS mutation type.

“Rather than treating KRAS as a single entity, this study reframes the field by asking a more precise question: ‘Which KRAS mutation?’ We show that different mutation types create distinct tumor ecosystems that can have real effects on patient outcomes,” said study leader Esra Akbay, Ph.D., Associate Professor of Pathology and a member of the Harold C. Simmons Comprehensive Cancer Center at UT Southwestern.
Up to a third of patients with lung adenocarcinoma, the most common form of lung cancer, have KRAS mutations that are thought to be responsible for tumor development and growth. About 41% of this group has a mutation type known as G12C, while 17% have a different mutation type known as G12D. These mutations were often assumed to drive cancer in the same ways, encouraging cells to become malignant, proliferate, and survive. However, tumors with G12C tend to respond better than those with G12D to a class of cancer drugs known as immune checkpoint inhibitors. The reason for this discrepancy has not been fully understood.
To find out why, Dr. Akbay and her colleagues worked with mice carrying these mutations. Cancer in those with the G12D mutation developed and progressed significantly faster than in those with the G12C mutation. Mice with the G12D mutation also survived for a significantly shorter time. The researchers reviewed large databases of cancer patients and found a similar scenario: Those with the G12D mutation tended to be diagnosed earlier in life, suggesting their tumors grow more rapidly.
When the researchers looked more closely at the tumors, they saw that genes associated with inflammation and immune activity were more active in mice carrying the G12C mutation. These tumors harbored more immune cells, including lymphocytes and cytotoxic T cells known to fight cancer, and more PDL1, the molecule targeted by immune checkpoint inhibitors. They also produced more antigens, molecules that signal the immune system.
Next, the researchers tested the effects of drugs that target each mutation type. While all the mice initially responded to these therapies, those with the G12D mutation relapsed faster than those with the G12C mutation. However, when mice with the G12D mutation were treated with a G12D-targeting drug, their tumors showed increased antigen presentation and PDL1 production and contained more immune cells, similar to those observed in G12C tumors. When the researchers administered an immune checkpoint inhibitor in addition to the G12D-inhibiting drug, many of the mice had a complete response, meaning that their tumors were totally eradicated.
Together, Dr. Akbay said, these findings suggest that KRAS mutation type matters, affecting the cancer cell behavior and immune activity of these cancers in different ways. Knowing a patient’s specific KRAS mutation could lead to personalized treatment plans incorporating mutation-specific inhibitors and immune checkpoint inhibitors that improve prognosis, she added.
Other UTSW researchers who contributed to this study are co-first authors Hai-Cheng Huang, Ph.D., doctoral student, and Qing Deng, Ph.D., postdoctoral researcher, both in the Akbay Lab; Luis De Las Casas, M.D., Professor of Pathology; Lin Xu, Ph.D., Assistant Professor in the Peter O’Donnell Jr. School of Public Health and of Pediatrics; and Lei Guo, Ph.D., Computational Biologist.
Dr. Akbay is a member of Simmons Cancer Center’s Development and Cancer Research Program.
This study was funded by grants from the Cancer Prevention and Research Institute of Texas (CPRIT Scholar Award RR160080), the National Institutes of Health (NIH) (R01CA289500, R01CA276058), National Cancer Institute (NCI) UT Southwestern-MD Anderson Cancer Center Specialized Program of Research Excellence (SPORE) (5P50CA070907), the American Cancer Society Research Scholar Award (RSG-22-051-01-IBCD), the Forbeck Foundation, and the NCI Cancer Center Support Grant (P30CA142543).
About UT Southwestern Medical Center
UT Southwestern, one of the nation’s premier academic medical centers, integrates pioneering biomedical research with exceptional clinical care and education. The institution’s faculty members have received six Nobel Prizes and include 24 members of the National Academy of Sciences, 25 members of the National Academy of Medicine, and 13 Howard Hughes Medical Institute Investigators. The full-time faculty of more than 3,300 is responsible for groundbreaking medical advances and is committed to translating science-driven research quickly to new clinical treatments. UT Southwestern physicians in more than 80 specialties care for more than 143,000 hospitalized patients, attend to more than 470,000 emergency room cases, and oversee nearly 5.3 million outpatient visits a year.
