Project Access & Support
Access for Small Projects
To gain access to the HTS facility and scientific staff for assay development and small screening projects, principal investigator(s) must first meet with the HTS Core Director, Bruce Posner, Ph.D., to discuss the primary assay, the goals for the screening project, assay development that has been completed to date, and downstream studies that will help advance the hits identified in the screen.
Access to the HTS Core is at the discretion of Dr. Posner and based largely on the originality of the screen, feasibility of the assay and screening strategy, and the therapeutic potential of the approach.
Once access is granted, the principal investigator will work collaboratively with the HTS lab to develop, optimize, and validate the assay for a small screen. Assay development is described in greater detail under HTS Screening of Chemical Compounds or siRNA Libraries.
Small projects are defined as screens that interrogate small chemical or RNAi libraries in the HTS Core or supplied by the principal investigator. The Core has several chemical sublibraries that include the Prestwick Collection (~1,100 approved drugs), the MacMillan Natural Products Fraction Collection (~7,360 fractions representing ~30,000 natural products), the NIH Clinical Collection (~450 experimental drugs), and the 2K subset (~1,800 natural product–like commercial compounds).
Access for Large Projects
In order to gain access to the HTS facility and the institutional libraries for large screening projects, principal investigators must submit a four-page proposal that includes the following elements:
- Abstract introducing the aims of the project and the approach to be used
- One page of background describing relevance of project to cancer
- Description of the screening strategy that will be employed
- Estimate of reagent, consumables, and instrument usage costs
- Name of scientist from the PI’s lab that will be dedicated to the experiment
- Description of secondary and counterscreening assays for prioritizing the compounds
- If applicable, a stated request for Cancer Center funding for screening supplies
Both the practicality and the scientific and therapeutic value of the approach for screening are assessed by the Cancer Center HTS Oversight Committee. The HTS team will help evaluate the cost and practicality of the proposed assay such as the number of liquid handling and wash steps, timing, reagent volumes, and necessary controls. If Cancer Center funding is requested, the HTS Oversight Committee will review the request and decide whether funds can be awarded. The maximal award is $12,500, and funds must be spent on screening supplies.
For advice about for applying for Cancer Center support, Bruce.Posner@UTSouthwestern.edu Dr. Posner.
To submit a proposal to the HTS Oversight Committee, send the proposal as an MS Word document or a PDF by Bruce.Posner@UTSouthwestern.edu to Dr. Posner. Dr. Posner will submit the proposal to the HTS Oversight Committee for review and provide feedback to the principal investigator.
Advanced Project Support
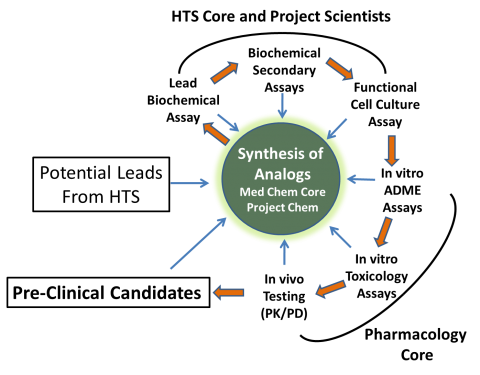
All data captured in each iteration of the lead optimization cycle is uploaded into the UT Southwestern Laboratory Information Management System (LIMS) to ensure the integrity of the data and facilitate data mining, reporting, and decision-making as leads are optimized and characterized. Drs. Anwu Zhou, Prema Latha Mallipeddi, and Hong Chen provide bio- and cheminformatic support for screening and serve as LIMS administrators. (For details on the LIMS, please see Core LIMS website.)