Study identifies transport protein key to immune response
UTSW researchers show how SEC24C moves STING to kick off signaling cascade, which could boost efforts to treat infections, cancer, other diseases
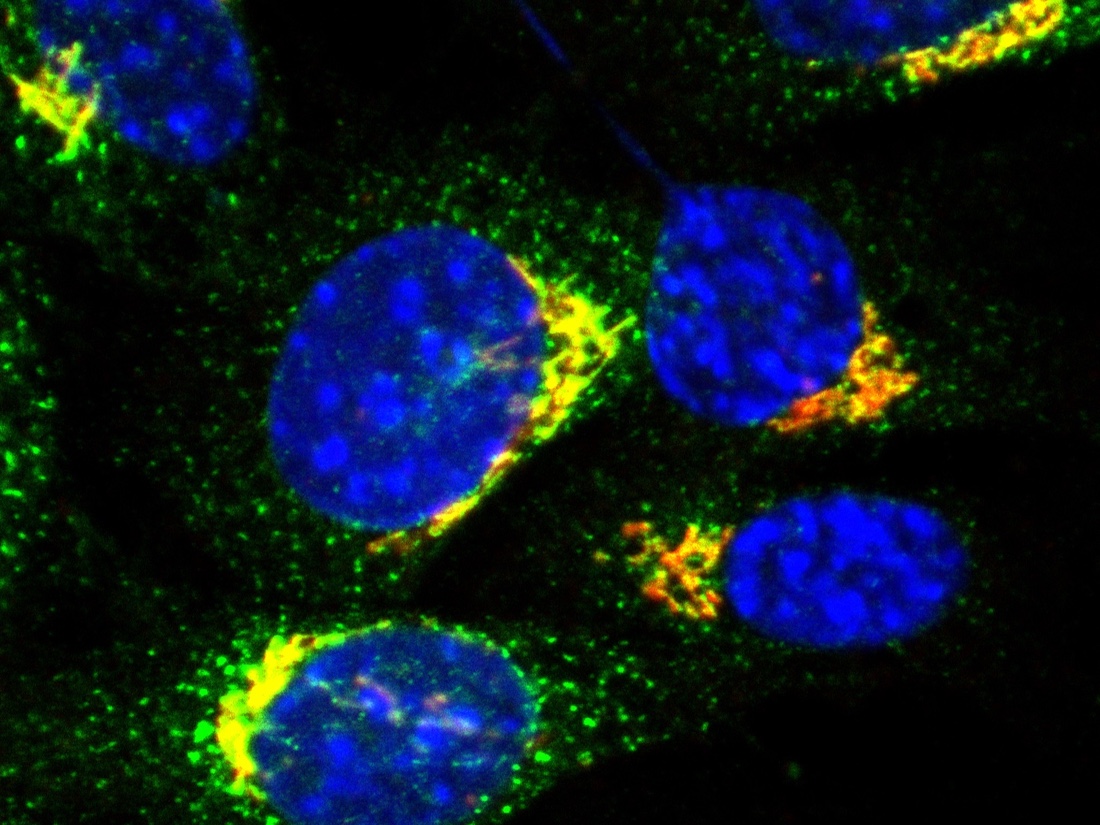
DALLAS – March 25, 2026 – UT Southwestern Medical Center researchers have identified how the quintessential immune protein known as stimulator of interferon genes (STING) migrates from one cellular organelle to another, a necessary step in its activation. The findings, reported in Cell, could eventually lead to new therapeutics that harness this system to fight infections, cancer, autoimmune disorders, and neurodegenerative diseases.
“Our study revealed structural insight on how STING achieves a controlled exit from the endoplasmic reticulum, which is essential for a balanced immune response,” said Nan Yan, Ph.D., Vice Chair and Professor of Immunology and Professor of Microbiology at UT Southwestern. Dr. Yan co-led the study with first author Heng Lyu, Ph.D., a postdoctoral researcher in the Yan Lab.

STING is a key part of the innate immune system, which provides broad and early protection against foreign invaders – such as viruses, bacteria, fungi, and parasites – as well as cancer. One trigger for innate immunity is DNA in the cytoplasm of cells. A protein called cGAS, discovered in 2012 by Zhijian “James” Chen, Ph.D., Professor of Molecular Biology and Director of the Center for Inflammation Research at UT Southwestern, senses this DNA. In response, it produces a molecule called cGAMP that binds to STING, which resides in a cellular organelle called the endoplasmic reticulum (ER) when it’s inactive.
After cGAMP binding, STING molecules are turned on, linking in a string (a process called oligomerization) and migrating to an organelle called the Golgi – a process the Yan Lab discovered in 2015. There, they activate additional molecules in a signaling cascade that prompts immune activity. STING’s oligomerization is necessary for this to occur.
Recent research led by Dr. Chen and colleagues, reported in two papers published concurrently in Nature, provided mechanistic and structural insight into how STING oligomerizes and why it needs to move from the ER to the Golgi. But how STING makes this migration and why oligomerization is critical to this process has been unclear.
To answer these questions, Dr. Yan and his team genetically engineered cells to delete four proteins that belong to a group called SEC24, which ferries proteins from the ER to other cellular locations. In those missing the protein known as SEC24C, STING could no longer initiate its immune signaling cascade when stimulated with a synthetic analog of cGAMP. These results suggest SEC24C moves STING from the ER to the Golgi.

To further confirm the interaction between these two proteins, the researchers used AlphaFold3 – a powerful artificial intelligence program that predicts the shape of proteins from their genetic sequence. They wanted to determine how SEC24C might bind to a pair of STING molecules attached together, the state in which STING exists in the ER. Their findings showed SEC24C appears to bind to STING in a region without a defined structure.
When the researchers mutated this region on STING, it no longer left the ER to migrate to the Golgi. The team had similar results when it mutated the region on SEC24C that AlphaFold3 predicted would bind to STING. These results suggested the disordered region on STING binds to a corresponding region on SEC24C to exit the ER.
Similarly, preventing STING from oligomerizing also stopped it from leaving the ER. A closer look showed that, unlike other proteins that SEC24C ferries from the ER to other cellular locations, STING’s disordered region is too short to bind strongly to the corresponding region on SEC24C. Thus, oligomerization is critical for creating a longer molecule that binds more strongly to SEC24C. This imperfect binding could be a way for cells to limit how easily STING is activated, Dr. Yan explained – an important protection against chronic STING activation that causes autoimmune disorders.
Further experiments showed that mutating the disordered region so it couldn’t bind to SEC24C impaired STING’s ability to fight off viral infections in cells. Conversely, mutations that increased binding strength to SEC24C boosted STING activity, helping it fight tumors in an animal cancer model.
Using drugs to tinker with STING’s binding to SEC24C could represent a new strategy for decreasing STING’s activity, possibly leading to treatments for autoimmune or neurodegenerative conditions, or for increasing it, potentially fighting infections or cancer. Dr. Yan and his colleagues plan to continue research toward this goal.
Other UTSW researchers who contributed to this study are Xuewu Zhang, Ph.D., Professor of Pharmacology and Biophysics; Wanwan Huai, Ph.D., Kun Song, Ph.D., and Hui Zhang, Ph.D., postdoctoral researchers; and Cong Xing, B.S., graduate student researcher.
Dr. Yan holds the Edwin L. Cox Distinguished Chair in Immunology and Genetics and is a Rita C. and William P. Clements, Jr. Scholar in Medical Research. Dr. Chen holds the George L. MacGregor Distinguished Chair in Biomedical Science. Drs. Yan and Chen are members of the Harold C. Simmons Comprehensive Cancer Center at UT Southwestern.
This study was funded by grants from the National Institutes of Health (AI151708 and R01CA273595), the Cancer Prevention and Research Institute of Texas (RP220242), and The Welch Foundation (I-1702).
About UT Southwestern Medical Center
UT Southwestern, one of the nation’s premier academic medical centers, integrates pioneering biomedical research with exceptional clinical care and education. The institution’s faculty members have received six Nobel Prizes and include 24 members of the National Academy of Sciences, 25 members of the National Academy of Medicine, and 13 Howard Hughes Medical Institute Investigators. The full-time faculty of more than 3,300 is responsible for groundbreaking medical advances and is committed to translating science-driven research quickly to new clinical treatments. UT Southwestern physicians in more than 80 specialties care for more than 143,000 hospitalized patients, attend to more than 470,000 emergency room cases, and oversee nearly 5.3 million outpatient visits a year.
