Studies identify lipids necessary for immune molecule activation
UTSW researchers’ discovery involving STING could open doors in fight against autoimmune disorders, infections, cancer, other diseases
DALLAS – Feb. 24, 2026 – UT Southwestern Medical Center researchers have identified two lipids that work together with a quintessential protein known as stimulator of interferon genes (STING) to launch an immune response in the human body. Their findings, detailed in two papers published concurrently in Nature, could lead to new ways to manipulate the immune system to fight infections, cancer, autoimmune disorders, and neurodegenerative diseases.
“These studies reveal additional levels of regulation of the cGAS-STING pathway, underscoring the importance of controlling the activity of this pathway so the body can mount an effective immune response against infections while avoiding autoimmune reactions to self-tissues. Dysregulation of this pathway has been shown to cause a variety of autoimmune and inflammatory diseases,” said Zhijian “James” Chen, Ph.D., Professor of Molecular Biology and Director of the Center for Inflammation Research at UT Southwestern.

Dr. Chen, one of the world’s leading researchers on innate immunity, is a co-author on one study and senior author on the other. His discovery of cGAS, an enzyme that produces a molecule called cGAMP to activate STING, has been recognized with numerous top honors including the 2026 Japan Prize in Life Sciences, the 2024 Albert Lasker Basic Medical Research Award, and the 2019 Breakthrough Prize in Life Sciences.
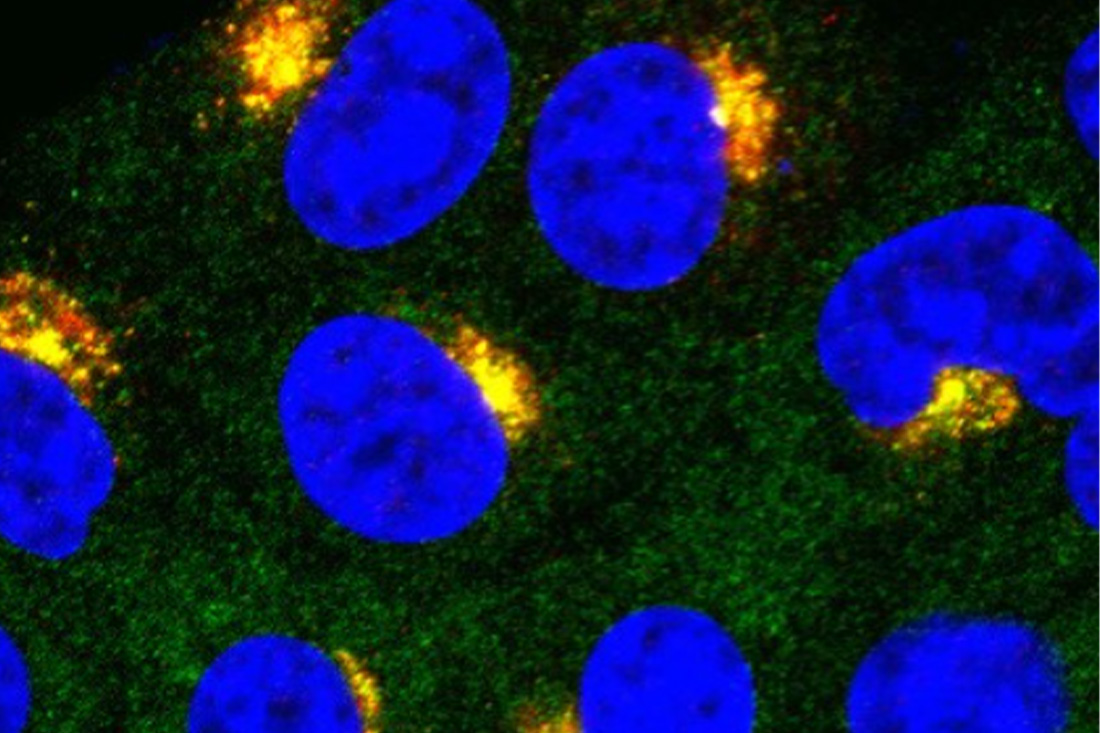
STING is a key part of the innate immune system, which provides broad and early protection against foreign invaders – such as viruses, bacteria, fungi, and parasites – as well as cancer. One trigger for innate immune activity is DNA found in the cytoplasm of cells. cGAS senses this DNA and produces cGAMP that binds to STING, which resides in a cellular organelle called the endoplasmic reticulum (ER) when it’s inactive.
After cGAMP binding, STING molecules are turned on, linking in a string (a process called oligomerization) and migrating to a different organelle called the Golgi. There, they activate additional molecules in a signaling cascade that prompts immune activity. STING’s oligomerization is necessary for this to occur.

How STING oligomerizes and why it needs to move from the ER to the Golgi have been unclear. To find additional molecules that play a role in STING regulation, a team led by Dr. Chen and Jay Xiaojun Tan, Ph.D., a former postdoctoral fellow in the Chen Lab who is now an Assistant Professor of Cell Biology at the University of Pittsburgh School of Medicine, analyzed which molecules interact with STING. One stood out: an enzyme known as PIKfyve, which produces a lipid called PtdIns(3,5)P2.
When the researchers used a genetic technique to delete PIKfyve from cells, STING no longer moved from the ER to the Golgi or activated molecules in its immune signaling cascade. Subsequent experiments showed that mixing PtdIns(3,5)P2 with STING enhanced STING activation by cGAMP. Further study showed that PtdIns(3,5)P2 directly binds to STING. Together, these results suggest that PtdIns(3,5)P2 works with cGAMP to activate STING.

Concurrently, Jie Li, Ph.D., Instructor of Biophysics at UTSW under the supervision of Xiaochen Bai, Ph.D., Associate Professor of Biophysics and Cell Biology, and Xuewu Zhang, Ph.D., Professor of Pharmacology and Biophysics, also were looking for molecules necessary to activate STING. Previous studies had shown that phosphatidylinositol phosphates (PIPs), the chemical family that includes PtdIns(3,5)P2, could play an important role in this process. Consequently, the researchers tested the effect of different PIPs on STING stimulated by cGAMP. Their experiments showed that PtdIns(3,5)P2 was necessary for STING molecules to link in a string.
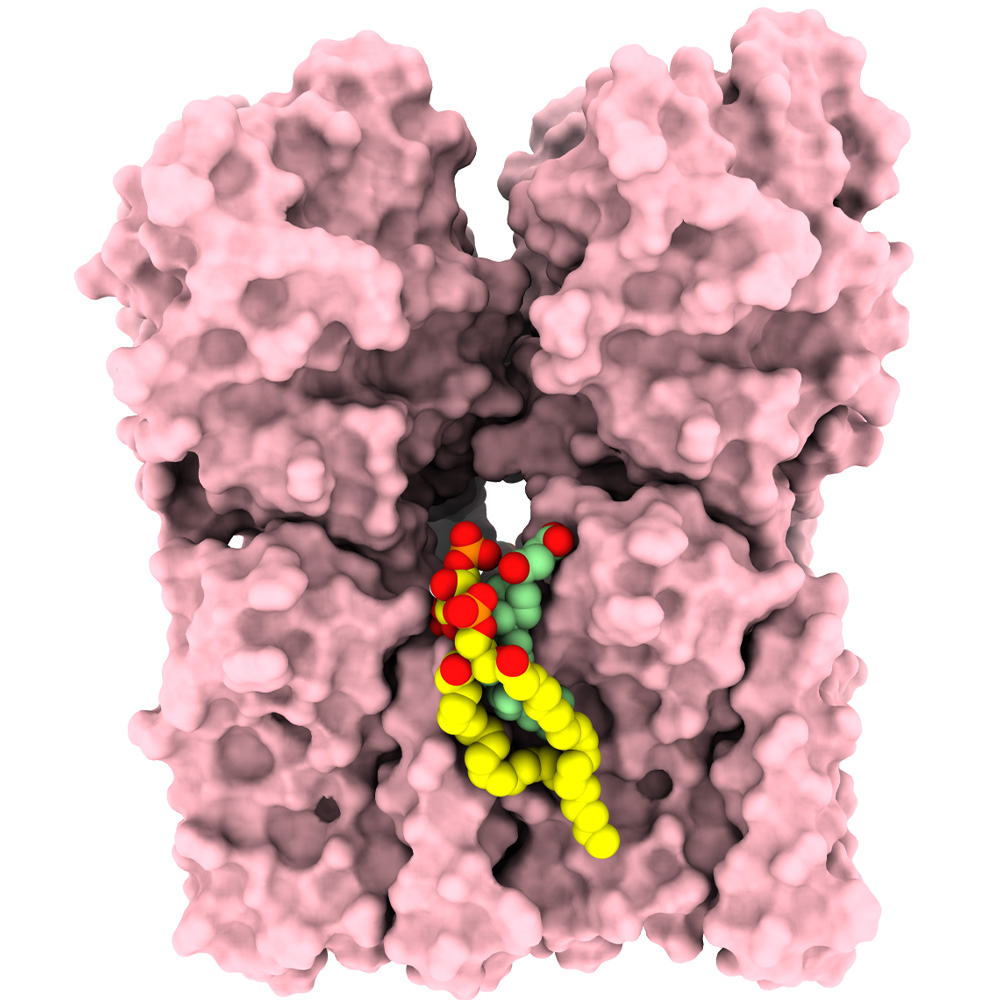
To better understand the role of PtdIns(3,5)P2, the researchers used cryo-electron microscopy, which can visualize molecules at the atomic level. Their findings showed that PtdIns(3,5)P2 binds to a groove between pairs of STING molecules, serving as a bridge that connects these pairs into a string. The researchers were surprised to also find cholesterol molecules in a place near where PtdIns(3,5)P2 binds. Cholesterol appears to stabilize STING’s linear arrangement.

Besides demonstrating PtdIns(3,5)P2’s key role in STING activation, these results also explain why STING must migrate from the ER to the Golgi to kick off its signaling cascade. While PtdIns(3,5)P2 and cholesterol are present in limited amounts in the ER, concentrations of these molecules are much higher in the Golgi and Golgi-derived vesicles – a necessary factor for STING to assemble into long chains.
Collaborating as a group, the two teams tested the effects of mutating parts of STING that bind to PtdIns(3,5)P2 and cholesterol. These mutant STING molecules no longer formed a chain or launched the signaling cascade, confirming that PtdIns(3,5)P2 and cholesterol are necessary for these events.
These findings don’t just answer basic science questions, Dr. Zhang explained. Rather, knowing the molecules involved in STING activation and how they bind to STING can guide researchers in designing drugs that can help or hinder these processes. Encouraging STING activation could help patients fight off infections or cancer, while stifling it could treat autoimmune and neurodegenerative diseases. The researchers plan to continue studying how the cGAS-STING pathway works to reach these goals.

Other UTSW researchers who contributed to the studies are Tuo Li, Ph.D., Assistant Professor of Molecular Biology; and Fenghe Du, M.D., and Xiang Chen, M.D., Research Specialists in the Chen lab.
Dr. Zhijian Chen, a member of both the National Academy of Sciences and the National Academy of Medicine, is also an Investigator of the Howard Hughes Medical Institute. He holds the George L. MacGregor Distinguished Chair in Biomedical Science. Dr. Zhang and Dr. Bai are Virginia Murchison Linthicum Scholars in Medical Research. Drs. Zhijian Chen, Bai, and Zhang are members of the Harold C. Simmons Comprehensive Cancer Center.
These studies were funded by grants from the National Institutes of Health (R01-AI093967, R01CA273595, and R01CA299257), The Welch Foundation (I-1389, I-1702, and I-1944), Cancer Grand Challenges (CGCFUL-2021/100007), Cancer Research UK, and the National Cancer Institute.
About UT Southwestern Medical Center
UT Southwestern, one of the nation’s premier academic medical centers, integrates pioneering biomedical research with exceptional clinical care and education. The institution’s faculty members have received six Nobel Prizes and include 24 members of the National Academy of Sciences, 25 members of the National Academy of Medicine, and 13 Howard Hughes Medical Institute Investigators. The full-time faculty of more than 3,300 is responsible for groundbreaking medical advances and is committed to translating science-driven research quickly to new clinical treatments. UT Southwestern physicians in more than 80 specialties care for more than 143,000 hospitalized patients, attend to more than 470,000 emergency room cases, and oversee nearly 5.3 million outpatient visits a year.
