Unusual kidney cancer feature sheds light into how cancers invade and metastasize
DALLAS – Oct. 12, 2021 – How cancers metastasize remains poorly understood. The process begins when cancer cells break off from a tumor and invade blood and lymphatic vessels, the body’s alleyways. But studying invasion - typically a microscopic process - is challenging. However, some types of tumors can invade blood vessels and form larger masses. By focusing on kidney cancer, a tumor that bulldozes its way into the largest veins, investigators at the Harold C. Simmons Comprehensive Cancer Center Kidney Cancer Program (KCP) at UT Southwestern now provide new insights. In a study published in Nature Communications, they show that invasion involves transient activation of an abnormal cell fate program, and that it is not always driven by the most evolved and aggressive cancer cell subsets (also called clones) in a tumor.

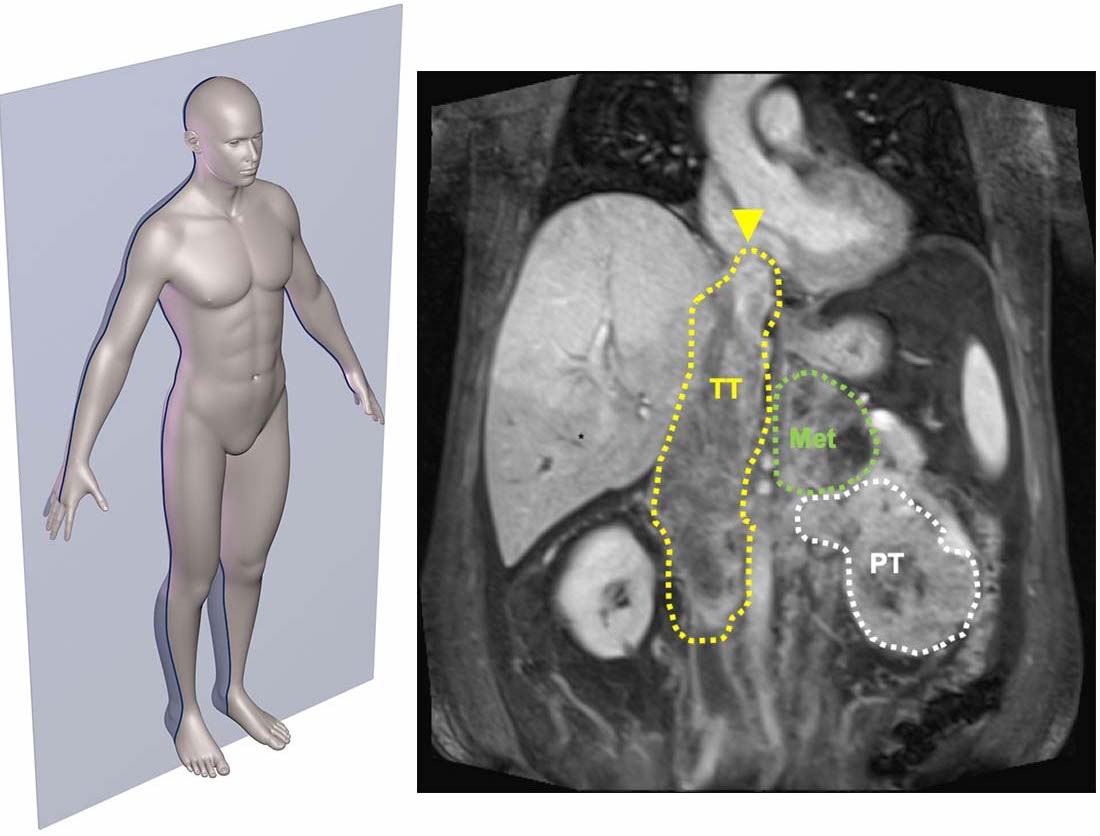
Among the top 10 cancers in the U.S., kidney cancer, also called renal cell carcinoma (RCC), has the unusual ability to use blood vessels as channels for direct expansion. In about 15 percent of patients, kidney cancer grossly invades into the renal vein from where it can reach the largest vein in the body, the inferior vena cava (IVC). From there, it has direct passage to the heart. When extending into the heart, the treatment is particularly challenging. This often requires sophisticated surgery with cardiac arrest and extracorporeal blood circulation using an artificial blood pump.
Assisted by KCP urologists, whose established track record treating kidney cancer draws referrals from across the region, researchers assembled a cohort of 83 patients who participated in the study. To understand invasion, they sampled multiple areas including from extensions into the vein and the tumor leading edge. By performing next-generation sequencing studies, they were able to dissect factors that contributed to invasion.
The results showed that invasion involves the transient activation of a gene program enabling cancer cells to survive within blood vessels. Unexpectedly, this was not always accomplished by the most aggressive cells in the tumor.
“Even less aggressive clones can invade and travel along the vein,” says study lead author, Srinivas Malladi, Ph.D., Assistant Professor of Pathology.
These data suggest that the ability to invade may be acquired earlier than previously thought. “It also explains why not all tumors that invade go on to metastasize,” says Malladi. “While some cells may have acquired the ability to invade, they have not yet gained the ability to survive in the blood stream or thrive in other organs.”
In clinical practice, the likelihood of metastases is determined based on the most evolved and aggressive clones within the tumor. However, KCP investigators found that metastases could be more accurately predicted based on the level of aggressiveness of the invading clones. After matching for aggressiveness in the tumor, patients with lesser aggressive tumor extensions had a four-fold lower risk of metastases.
“This suggests,” says study co-leader Payal Kapur, M.D., Professor of Pathology and Urology and Group Leader of Genitourinary Pathology, “that we should pay more attention to grading tumor extensions, as this may give patients a more accurate assessment of their likelihood to develop metastases.” She adds, “If validated, this could change current practice.”
Thus far, treatment advances have largely focused on improving surgical techniques, but Malladi hopes that the study will help. “By blocking the cell fate program, the so called EMT program, we might be able to prevent tumor cells from invading.”
The study results are the latest in a series of pioneering research and treatment advancements for patients with this type of invasive tumors. “This is a patient population that hasn’t seen substantial improvements in outcomes for many years,” says study co-leader James Brugarolas, M.D., Ph.D., Professor of Internal Medicine, Hematology/Oncology, and KCP Director.
Surgery is fraught with risks and the potential for life-threatening complications. Without treatment, patients whose tumors invade the IVC often die within a year.
Looking for better options, especially for patients who are not good candidates for surgery, KCP investigators have developed a novel approach involving radiation. While the treatment remains experimental, their data suggest that large doses of precisely delivered radiation can control tumor extensions.
“These are important advances,” says Brugarolas. “The KCP is invested in helping these patients by improving surgical techniques and developing new treatment approaches, as well as by achieving a deeper understanding of the underlying biological process.”
Dr. Brugarolas holds the Sherry Wigley Crow Cancer Research Endowed Chair in Internal Medicine, in honor of Robert Lewis Kirby, M.D.
Dr. Kapur holds the Brock Fund for Medical Science and the Jan and Bob Pickens Distinguished Professorship in Medical Science, in honor of Jerry Knight Rymer and Annette Brannon Rymer and Mr. and Mrs. W.L. Pickens.
UT Southwestern co-authors include: Kangsan Kim, Qinbo Zhou, Alana Christie, Christina Stevens, Yuanqing Ma, Oreoluwa Onabolu, Suneetha Chintalapati, Tiffani Mckenzie, Vanina Toffessi Tcheuyap, Layton Woolford, Zhiqun Xie, He Zhang, Nirmish Singla, Pravat Kumar Parida, Mauricio Marquez-Palencia, Ivan Pedrosa, Vitaly Margulis, Arthur Sagalowsky, Tao Wang, Zora Modrusan, Somasekar Seshagiri, Payal Kapur.
This work was supported by grants from CPRIT (RP190208, RP180192, RP180191, RR170003), NCI (5R01CA154475), NIH (P50CA196516, R01DK115986), DOD (W81XWH1910710), NSF (2019281049), ACS (RSG-20-47-01-CSM), and Cancer Center Support (P30 CA142543) grants.
The authors declare no competing interests.
UT Southwestern’s Harold C. Simmons Comprehensive Cancer Center, the only National Cancer Institute-designated Comprehensive Cancer Center in North Texas, includes five research and 12 clinical care programs with a focus on fostering groundbreaking translational research that can improve patient treatment, address cancer health disparities, and prevent cancer worldwide. In addition, the Center’s education and training programs support and develop the next generation of cancer researchers and clinicians. The comprehensive designation and associated funding is designed to bolster the Simmons Cancer Center’s research and to provide patients access to innovative clinical trials with promising new drugs. Simmons Cancer Center members currently have over $90 million in extramural cancer-focused research funding.
About UT Southwestern Medical Center
UT Southwestern, one of the nation’s premier academic medical centers, integrates pioneering biomedical research with exceptional clinical care and education. The institution’s faculty members have received six Nobel Prizes and include 24 members of the National Academy of Sciences, 25 members of the National Academy of Medicine, and 13 Howard Hughes Medical Institute Investigators. The full-time faculty of more than 3,300 is responsible for groundbreaking medical advances and is committed to translating science-driven research quickly to new clinical treatments. UT Southwestern physicians in more than 80 specialties care for more than 143,000 hospitalized patients, attend to more than 470,000 emergency room cases, and oversee nearly 5.3 million outpatient visits a year.
