Regenerating hope for spinal cord injuries
In a stunning advance in the field of stem cell research, UT Southwestern scientists successfully boosted the regeneration of mature nerve cells in the spinal cords of adult mammals by tenfold.

“This research lays the groundwork for regenerative medicine for spinal cord injuries. We have uncovered critical molecular and cellular checkpoints in a pathway involved in the regeneration process that may be manipulated to boost nerve cell regeneration after a spinal injury,” said Dr. Chun-Li Zhang, Associate Professor of Molecular Biology, a member of the Hamon Center for Regenerative Science and Medicine, and a W.W. Caruth, Jr. Scholar in Biomedical Research.
Dr. Zhang, senior author of a study outlining the findings in Cell Reports, cautioned that this research in mice is still in the early experimental stage and is not ready for clinical translation.
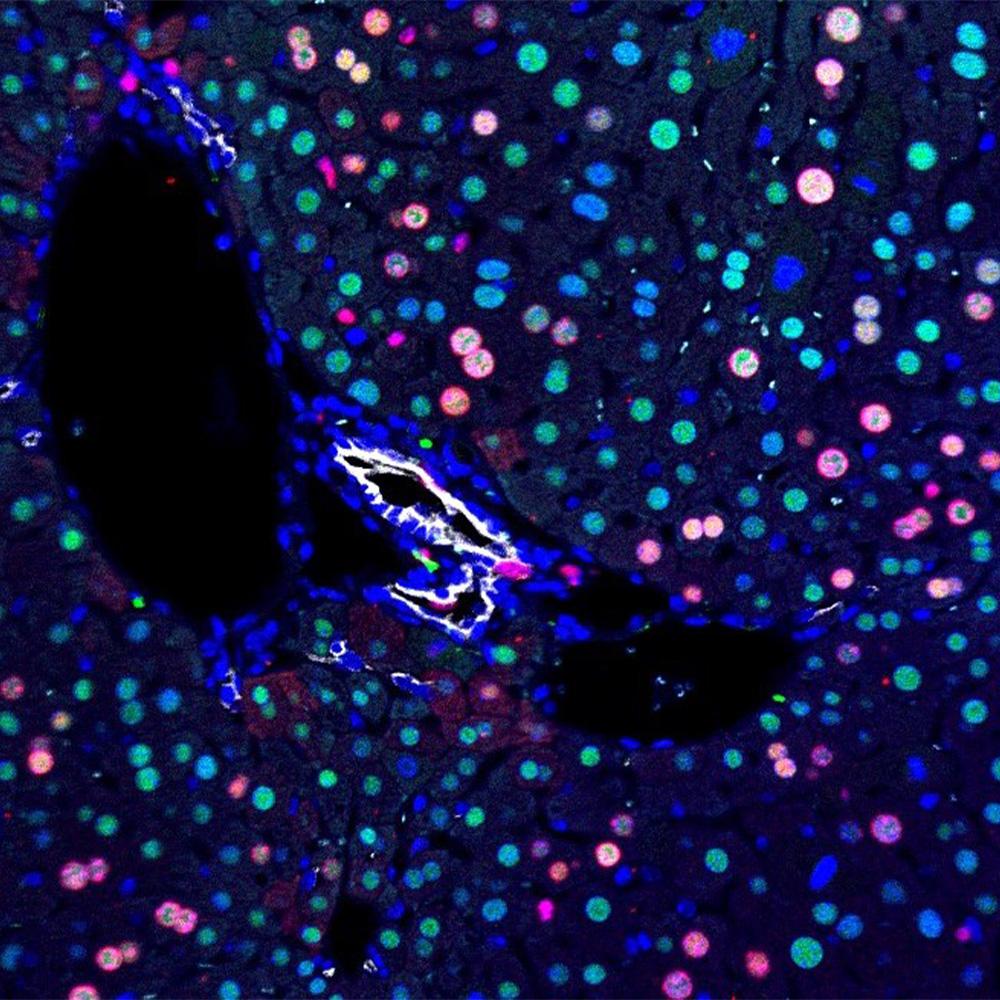
A series of screens in living animals led to a two-step process that overcame the propensity of glial cells – the most common cells in the spinal cord – to form scar tissue following injury. Study lead author Dr. Lei-Lei Wang, a postdoctoral researcher in Dr. Zhang’s lab, led this effort.
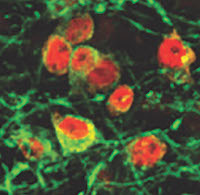
The researchers first silenced parts of the p53-p21 protein pathway that acts as a roadblock to the reprogramming of glial cells into the more primitive, stem-like types of cells that have the potential to become nerve cells. In the second step, mice were screened for factors that could boost the number of stem-like cells that matured into adult neurons. Two growth factors – BDNF and Noggin – accomplished this goal.
This work built on earlier research at UT Southwestern in 2013 and 2014, when scientists in the Zhang lab created new nerve cells in the brains and spinal cords of mice by introducing transcription factors that promoted the transition of adult glial cells into more primitive, stem cell-like states, then coaxed them to mature into adult nerve cells.