Study details how general anesthetics and 'benzos' act on receptors in the brain
Understanding how the anesthetics and psychoactive drugs work at a molecular level could eventually lead to new, more targeted drugs with fewer side effects

DALLAS – Sept. 2, 2020 – As you drift into unconsciousness before a surgery, general anesthetic drugs flowing through your blood are putting you to sleep by binding mainly to a protein in the brain called the ɣ-aminobutyric acid type A (GABAA) receptor. Now UT Southwestern scientists have shown exactly how anesthetics attach to the GABAA receptor and alter its three-dimensional structure, and how the brain can tell the difference between anesthetics and the psychoactive drugs known as benzodiazepines – which also bind to the GABAA receptor. The findings were published online today in the journal Nature.
“Anesthetics remain one of the most clinically important, yet mysterious, classes of drugs,” says study leader Ryan Hibbs, Ph.D., an associate professor of neuroscience and biophysics at UTSW and senior author of the new paper. “We went into this study driven by curiosity about how general anesthesia works – and now we’re one big step closer to answering that question.”
General anesthetics, which can be inhaled or administered intravenously, put people into a sleep-like, immobilized state. Although intravenous anesthetics have been used since the 1930s, it wasn’t until around the turn of the 21st century that researchers discovered that the drugs act on the GABAA receptor in the brain.
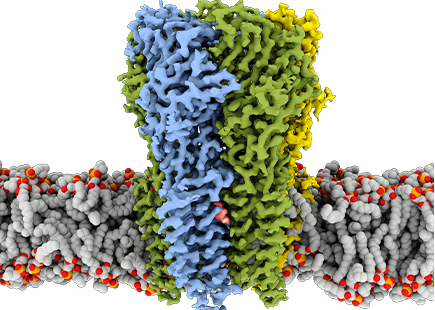
The GABAA receptor is an ion channel; when it’s in an open conformation, it allows chloride ions to flow through. This movement of ions decreases the signaling of brain cells, calming brain activity. So stimulating the GABAA receptor – as anesthetics, benzodiazepines, alcohol, anti-seizure, and some sleep medications all do – quiets the brain in a variety of ways.
In 2018, Hibbs’ lab group detailed the first ever atomic structure of the GABAA receptor. In the new study, Hibbs and his colleagues looked again at the structure of the receptor in an environment more closely mimicking the cell, and this time while it was being bound by one of three different anesthetics – phenobarbital, etomidate, and propofol – as well as the benzodiazepine drug diazepam, or Valium, which is used to treat anxiety disorders, and the drug flumazenil, which can treat benzodiazepine overdoses.
“What we found is that the GABAA receptor is particularly sensitive to its surrounding environment,” says Jeong Joo Kim, Ph.D., a UTSW postdoctoral research fellow and first author of the study. “It has the ability to change conformation in different ways based on the binding of many different drugs.”
The team discovered that both general anesthetics and diazepam could bind to multiple places on the GABAA molecule. One site – dubbed the “benzo site” in earlier research – was unique to the diazepam. But another site overlapped between the two drug types. When diazepam was present at high enough doses, it bound to this site that was more often used by the anesthetics. This observation could explain why high doses of benzodiazepines like diazepam can have anesthetic-like effects. The researchers also found differences among the general anesthetics; phenobarbital, for instance, bound to a place on GABAA that neither etomidate nor propofol attached, and seemed to be less choosy about where it bound.
The nuances between how and where each drug binds to the GABAA receptor raise the possibility of engineering new drugs that could be more selective for certain effects on the brain or have fewer side effects.
“The fact that there are differences in the binding sites gives us some hope that we might be able to create more specific molecules that bind to only one site on the GABAA receptor,” says Hibbs, an Effie Marie Cain Scholar in Medical Research. “This is now a launching point for the discovery of improved, more selective anesthetics.”
In addition to the observations on exactly which part of the GABAA receptor the drugs bound to, the researchers also studied how the conformation of the rest of the GABAA molecule was different in response to each drug. The anesthetics, they found, stabilized a version of the GABAA receptor that opened – to allow ions through – much more easily than usual. This leads to the dramatic calming of the brain required for general anesthesia. Diazepam stabilized a receptor structure that was intermediate – the channel opened more easily than normal, but wasn’t as open as with the anesthetics. Such an intermediate structure likely allows some of the benefits of lower dose benzodiazepines to treat anxiety, epilepsy, and insomnia without causing unconsciousness.
Hibbs and his lab plan to conduct similar structural studies for other drugs that are known to bind to GABAA, including Ambien (zolpidem) and a class of neurosteroids used to treat epilepsy.
“This is just a foundation,” he says. “By continuing to study other classes of drugs that interact with this receptor, we can get an even more complete three-dimensional blueprint of the GABAA receptor and its pharmacology.” That insight, he adds, may help shed light on not only how we can tweak the activity of the GABAA receptor for medical gains, but explore the very nature of human consciousness.
Other UTSW researchers who contributed to this study were Anant Gharpure, Jinfeng Teng, Shaotong Zhu, and Colleen Noviello. Collaborators were Yuxuan Zhuang, Rebecca Howard, and Erik Lindahl of Stockholm University and Richard Walsh of Harvard Medical School.
This research was supported by funds from the American Heart Institute 20POST35200127 and 18POST34030412, Vetenskapsrådet VR, the Knut and Alice Wallenberg Foundation, The Welch Foundation (I-1812), and the National Institutes of Health DA037492, DA042072, and NS095899.
About UT Southwestern Medical Center
UT Southwestern, one of the nation’s premier academic medical centers, integrates pioneering biomedical research with exceptional clinical care and education. The institution’s faculty members have received six Nobel Prizes and include 24 members of the National Academy of Sciences, 25 members of the National Academy of Medicine, and 13 Howard Hughes Medical Institute Investigators. The full-time faculty of more than 3,300 is responsible for groundbreaking medical advances and is committed to translating science-driven research quickly to new clinical treatments. UT Southwestern physicians in more than 80 specialties care for more than 143,000 hospitalized patients, attend to more than 470,000 emergency room cases, and oversee nearly 5.3 million outpatient visits a year.
