Surveillance pathway tells cells when they run low on lipids
UT Southwestern study could lead to new treatments for metabolic disorders and other health conditions

DALLAS – May 18, 2022 – UT Southwestern researchers have discovered a molecular pathway that allows cells to sense when their lipid supplies become depleted, prompting a flurry of activity that prevents starvation. The findings, reported in Nature, might someday lead to new ways to combat metabolic disorders and a variety of other health conditions.
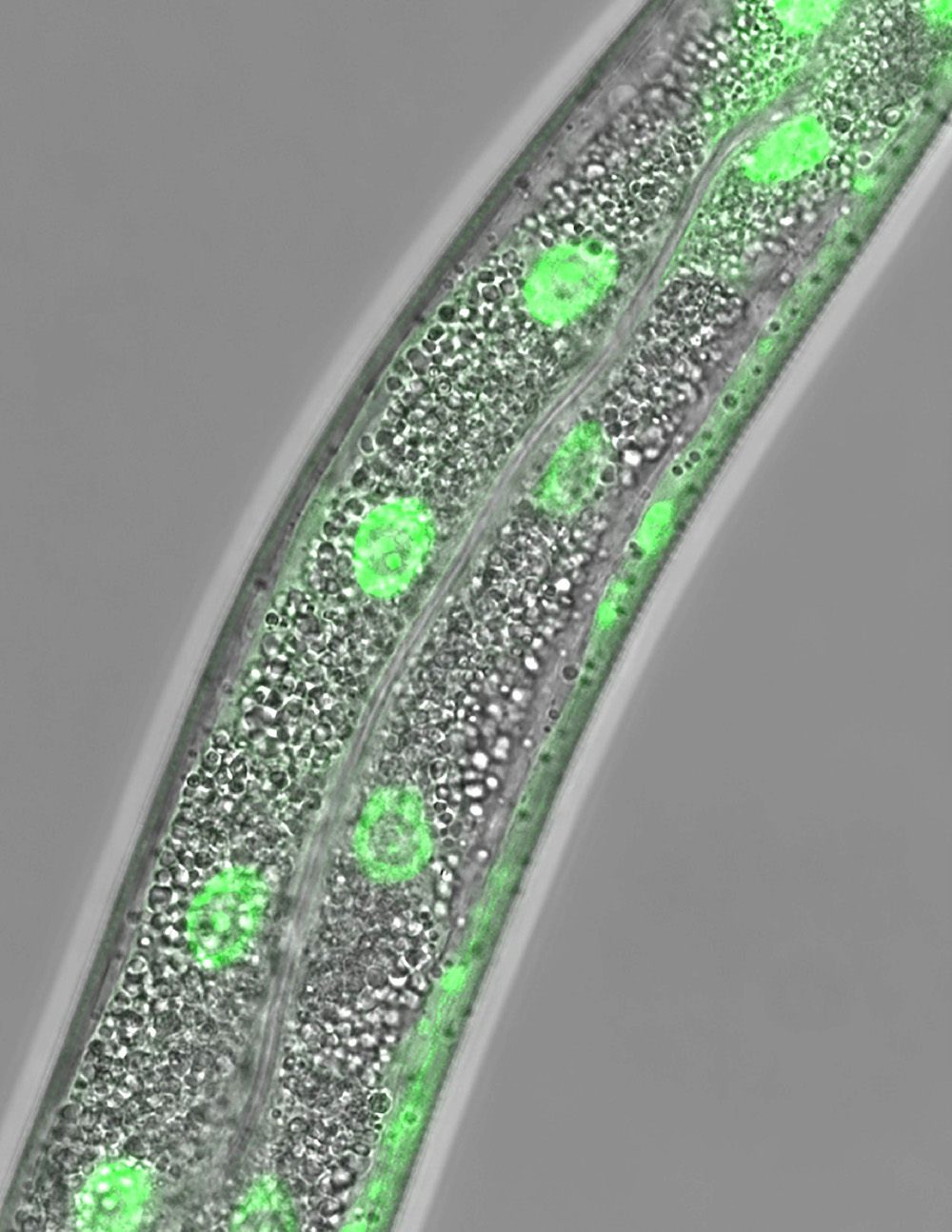
“Lipids are critical for supplying energy and serving as components for membranes and other cellular structures,” said study leader Peter Douglas, Ph.D., Assistant Professor of Molecular Biology at UT Southwestern. “The mechanism we discovered appears to be a way for cells to universally gauge lipid levels without having to distinguish between the vast variety of different lipid types.”
Dr. Douglas, a member of the Hamon Center for Regenerative Science and Medicine at UT Southwestern, explained that mammalian cells possess tens of thousands of different types of chemically distinct lipids, a class of molecules composed of hydrocarbon chains that are the most energy-rich chemicals in the body. In the 1990s, UT Southwestern Nobel Prize winners Michael Brown, M.D., and Joseph Goldstein, M.D., both Professors of Molecular Genetics and Internal Medicine, discovered the first lipid-sensing pathway: the SREBP pathway, which helps cells regulate cholesterol levels by sensing a type of lipids called sterols.
Components in the SREBP pathway perform this feat by directly binding sterol molecules. However, to accommodate the sheer number of different lipid types, Dr. Douglas said additional sensing mechanisms were likely to exist in the cell.
Searching for a mechanism that cells might generally apply to sense lipid levels, the researchers starved Caenorhabditis elegans, a roundworm species that serves as a common lab model and shares many genes with humans. When these animals were deprived of food for 24 hours, the researchers saw that a protein known as nuclear hormone receptor 49 (NHR-49) moved from the cytosol – the liquid component of cells – to the nucleus, where it set off a cascade of gene activity that prompted transport of other proteins to the cell surface to gather extracellular nutrients.
The researchers found that when C. elegans were well fed, NHR-49 was held in place by a protein known as RAB-11.1. This protein falls into a class known as small G proteins that were discovered by the late Alfred G. Gilman, M.D., Ph.D., a Nobel Laureate and longtime Chair of Pharmacology at UTSW who later served as Dean of the Medical School, Executive Vice President for Academic Affairs, and Provost. But under starvation conditions, RAB-11.1 released NHR-49, allowing its nuclear entry.
Further experiments showed that this release is triggered by the lack of a lipid known as geranylgeranyl pyrophosphate. Because cells can use almost any lipid as a starting material to make geranylgeranyl pyrophosphate, its presence served as a universal signal for acceptable lipid levels in cells, Dr. Douglas explained.
He added that prior research by other labs has shown that components of this pathway are active in a number of diseases and health problems, including metabolic disease, cardiovascular disease, viral infections, neurological aging, and some cancers. Manipulating this new lipid-sensing pathway could eventually lead to new treatments for these conditions, Dr. Douglas said.
Dr. Douglas is a Southwestern Medical Foundation Scholar in Biomedical Research and a Cancer Prevention and Research Institute of Texas (CPRIT) Scholar.
Other UT Southwestern researchers who contributed to this study include Abigail Watterson, Lexus Tatge, Naureen Wajahat, Sonja L.B. Arneaud, Rene S. Fonseca, Shaghayegh T. Beheshti, Patrick Metang, Melina Mihelakis, Kielen R. Zuurbier, Chase D. Corley, Ishmael Dehghan, and Jeffrey G. McDonald.
This work was funded by the Clayton Foundation for Research, The Welch Foundation (I-2061-20210327), the National Institutes of Health (R00AG042495, R01AG061338, and R56AG070167), and CPRIT (RR150089).
Drs. Brown and Goldstein are Regental Professors at UTSW. Dr. Goldstein is Chair of Molecular Genetics and Dr. Brown is Director of the Erik Jonsson Center for Research in Molecular Genetics and Human Disease. Dr. Brown holds the W. A. (Monty) Moncrief Distinguished Chair in Cholesterol and Arteriosclerosis Research, and the Paul J. Thomas Chair in Medicine. Dr. Goldstein holds the Julie and Louis A. Beecherl, Jr. Distinguished Chair in Biomedical Research, and the Paul J. Thomas Chair in Medicine.
About UT Southwestern Medical Center
UT Southwestern, one of the nation’s premier academic medical centers, integrates pioneering biomedical research with exceptional clinical care and education. The institution’s faculty members have received six Nobel Prizes and include 24 members of the National Academy of Sciences, 25 members of the National Academy of Medicine, and 13 Howard Hughes Medical Institute Investigators. The full-time faculty of more than 3,300 is responsible for groundbreaking medical advances and is committed to translating science-driven research quickly to new clinical treatments. UT Southwestern physicians in more than 80 specialties care for more than 143,000 hospitalized patients, attend to more than 470,000 emergency room cases, and oversee nearly 5.3 million outpatient visits a year.
