Scientists identify cells responsible for liver tissue maintenance and regeneration

DALLAS – Feb. 25, 2021 – While the amazing regenerative power of the liver has been known since ancient times, the cells responsible for maintaining and replenishing the liver have remained a mystery. Now, research from the Children’s Medical Center Research Institute at UT Southwestern (CRI) has identified the cells responsible for liver maintenance and regeneration while also pinpointing where they reside in the liver.
These findings, reported today in Science, could help scientists answer important questions about liver maintenance, liver damage (such as from fatty liver or alcoholic liver disease), and liver cancer.
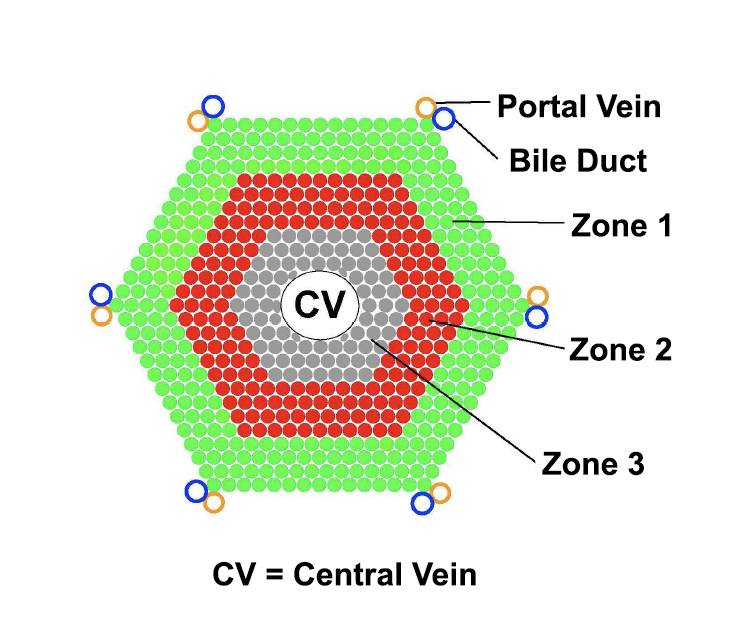
The liver performs vital functions, including chemical detoxification, blood protein production, bile excretion, and regulation of energy metabolism. Structurally, the liver is comprised of tissue units called lobules that, when cross-sectioned, resemble honeycombs. Individual lobules are organized in concentric zones in which hepatocytes, the primary liver cell type, carry out diverse functions. Over the past 10 years, there has been debate about whether all hepatocytes across the lobule contribute to the production of new cells or if a certain subset of hepatocytes or stem cells is responsible.

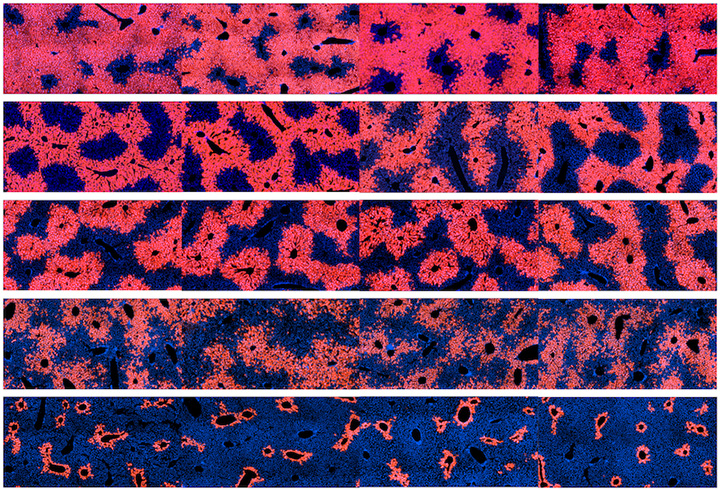
Previous efforts to identify the cells most responsible for liver regeneration were hindered by a lack of markers to distinguish and compare the functions of distinct types of hepatocytes in different regions of the liver. Scientists in the Zhu lab addressed this issue by comparing the genes that mark hepatocytes throughout the liver. Using this approach, they identified genes that were only turned on by specific subsets of hepatocytes, and then used these genes as markers to distinguish the identities and functions of different hepatocyte subsets. They created 11 new mouse strains, each of which carries a marker for a specific subset of hepatocytes. Along with three previously established mouse strains, researchers observed how the labeled cells multiplied or disappeared over time, and which were responsible for liver regeneration after damage. These experiments allowed researchers to directly compare how different subsets of hepatocytes contributed to liver maintenance and regeneration.
Members of the Zhu lab discovered that cells in zone 2 gave rise to new hepatocytes that populated all three zones of liver lobules while cells from zones 1 and 3 disappeared. These unexpected observations suggested that there is not a rare population of stem cells responsible for liver maintenance, but instead, a common set of mature hepatocytes within a specific region of the liver that regularly divide to make new hepatocytes throughout the liver. The Zhu lab also exposed mice to chemicals that mimicked common forms of liver damage, showing that cells in zone 2 were most able to evade death, regenerate hepatocytes, and sustain liver function.
“In humans, cells in zones 1 and 3 are most often harmed by alcohol, acetaminophen, and viral hepatitis. So it makes sense that cells in zone 2, which are sheltered from toxic injuries affecting either end of the lobule, would be in a prime position to regenerate the liver. However, more investigation is needed to understand the different cell types in the human liver,” says Hao Zhu, M.D., an associate professor at CRI and lead author of the study.
To learn more about mechanisms that hepatocytes in zone 2 use to regenerate liver function, members of the Zhu lab performed genetic screens to look for genes important for growth and regeneration. They discovered a pathway known as the IGFBP2-mTOR-CCND1 axis that was active in zone 2 but less so in zones 1 and 3. When they deleted components of this pathway from mice, the cells in zone 2 no longer gave rise to new hepatocytes, establishing that this was the mechanism responsible for the regenerative capacity of zone 2 cells.
“The identification of zone 2 hepatocytes as a regenerative population answers some fundamental questions about liver biology and could have important implications for liver disease. In addition, the tools we created to study different types of hepatocytes can be used to examine how different cells respond to liver damage or to genetic changes that cause liver cancer,” says Zhu.
Zhu is an associate professor of pediatrics and internal medicine at UT Southwestern where he holds the Kern Wildenthal, M.D., Ph.D. Distinguished Professorship in Pediatric Research. He is also a Cancer Prevention and Research Institute of Texas (CPRIT) Scholar in Cancer Research.
The Pollack Foundation, National Institutes of Health (NIH)/National Institute of Diabetes and Digestive and Kidney Diseases (R01DK111588), NIH/National Cancer Institute (R01CA251928), CPRIT (RP170267, RP180268), a Burroughs Wellcome Fund Career Award for Medical Scientists, a Stand Up To Cancer Innovative Research Grant (SU2C-AACR-IRG 10-16), and donors to the Children’s Medical Center Foundation supported this work.
About CRI
Children’s Medical Center Research Institute at UT Southwestern (CRI) is a joint venture of UT Southwestern Medical Center and Children’s Medical Center Dallas. CRI’s mission is to perform transformative biomedical research to better understand the biological basis of disease. Located in Dallas, Texas, CRI is home to interdisciplinary groups of scientists and physicians pursuing research at the interface of regenerative medicine, cancer biology, and metabolism.
