New gene editing strategies developed for Duchenne muscular dystrophy
DALLAS – April 30, 2021 – UT Southwestern scientists successfully employed a new type of gene therapy to treat mice with Duchenne muscular dystrophy (DMD), uniquely utilizing CRISPR-Cas9-based tools to restore a large section of the dystrophin protein that is missing in many DMD patients. The approach, described online today in the journal Science Advances, could lead to a treatment for DMD and inform the treatment of other inherited diseases.

“Thousands of different mutations causing Duchenne have been identified, but they tend to cluster into certain parts of the dystrophin gene,” says study leader Eric Olson, Ph.D., professor and founding chair of molecular biology at UTSW. Some of these mutations cause muscle cells to produce short, less functional versions of the dystrophin protein. “The power of our method is that you don’t need a new gene editing strategy for every patient with a new mutation; you can correct multiple different mutations with a consolidated approach.”
Olson and his colleagues took advantage of the fact that the enormous dystrophin gene is composed of many different segments, called exons, some of which are dispensable. In about 8 percent of boys with DMD, nearly half of the dystrophin protein is missing due to mutations within exon 51 that cause the body to stop protein production. The researchers developed multiple successful CRISPR-Cas9 nucleotide gene editing strategies to skip the errant “stop” signal, restoring production of 97 percent of the protein. Some strategies worked by removing neighboring exons, while others utilized tiny genetic additions or subtractions to get protein production back on track.
When the researchers used the new approach in mice with dystrophin mutations, functioning copies of dystrophin returned to more than half of all leg muscle fibers within three weeks. Moreover, the group showed that they could use isolated cells from mice or humans with DMD to test whether the approach would be successful for a particular patient ahead of treatment. The isolated cells are coaxed to develop into induced pluripotent stem cells (iPS cells) and then heart cells. In a dish, researchers can observe whether the gene editing program helps the heart cells work better.
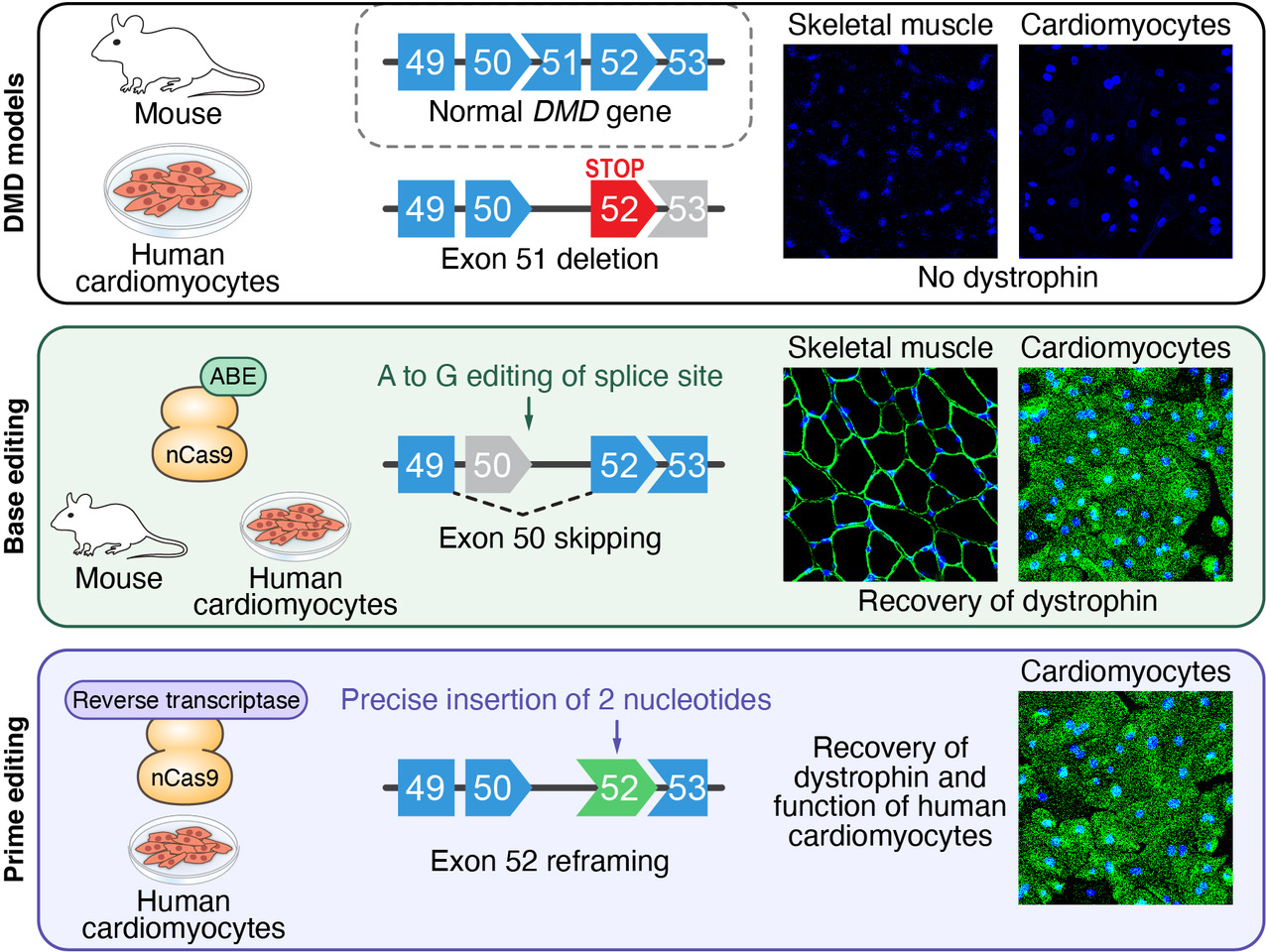
“Using iPS cell-derived cardiomyocytes from DMD patients, we rapidly tested our nucleotide gene editing approaches, demonstrating the recovery of the dystrophin protein,” says Francesco Chemello, Ph.D., a postdoctoral researcher in the Olson lab and first author of the paper.
DMD affects about one in five thousand males at birth and leads to progressively worsening muscle weakness in early childhood. The disease is caused by one of more than 7,000 different mutations in the gene for dystrophin – a protein that normally acts as a scaffold to support muscle fibers. Without fully functional dystrophin, the skeletal and heart muscles of people with DMD degenerate over time, eventually leading to death.
The gene therapy reported in the study is not ready for humans with DMD yet. Additional safety studies in animals are needed first, as well as more work to optimize the virus that carries the gene editing machinery to muscle and heart cells. But by showing that multiple CRISPR-Cas9 strategies can correct a mutation, the researchers have expanded the toolbox of potential gene therapy options for DMD.
“Every cell in the human body has 3 billion letters of DNA sequence in its genome, and this method makes it possible to correct large deletions in the DMD gene by specifically swapping one of these letters,” says Olson. “That level of specificity and efficiency is remarkable.”
Olson holds The Robert A. Welch Distinguished Chair in Science, the Pogue Distinguished Chair in Research on Cardiac Birth Defects, and the Annie and Willie Nelson Professorship in Stem Cell Research.
Other UTSW researchers who contributed to this study are Andreas C. Chai, Hui Li, Cristina Rodriguez-Caycedo, Efrain Sanchez-Ortiz, Ayhan Atmanli, Alex A. Mireault, Ning Liu and Rhonda Bassel-Duby.
This research was supported by funds from the National Institutes of Health (HL-130253, AR-071980, and AR-067294), the Senator Paul D. Wellstone Muscular Dystrophy Specialized Research Center (HD-087351), Cancer Prevention and Research Institute of Texas (RP200103), and the Robert A. Welch Foundation (grant 1-0025).
About UT Southwestern Medical Center
UT Southwestern, one of the nation’s premier academic medical centers, integrates pioneering biomedical research with exceptional clinical care and education. The institution’s faculty members have received six Nobel Prizes and include 24 members of the National Academy of Sciences, 25 members of the National Academy of Medicine, and 13 Howard Hughes Medical Institute Investigators. The full-time faculty of more than 3,300 is responsible for groundbreaking medical advances and is committed to translating science-driven research quickly to new clinical treatments. UT Southwestern physicians in more than 80 specialties care for more than 143,000 hospitalized patients, attend to more than 470,000 emergency room cases, and oversee nearly 5.3 million outpatient visits a year.
