Gene in fat plays key role in insulin resistance
Developing a better understanding of KBTBD2 could have implications for Type 2 diabetes, UTSW researchers say
DALLAS – July 24, 2020 – Deleting a key gene in mice in just their fat made tissues throughout these animals insulin resistant, in addition to other effects, a new study by UT Southwestern researchers shows. The findings, reported in a recent issue of PNAS, could shed light on Type 2 diabetes and other insulin resistance disorders, which remain poorly understood despite decades of study.

Photo taken by Brian Coats for UT Southwestern Medical Center
In 2016, UTSW immunologist and geneticist Bruce Beutler, M.D., Zhao Zhang, Ph.D., and their colleagues reported a new mouse mutant that they named teeny, which resulted from inactivating a gene known as KBTBD2 that is widely expressed throughout the body in mice and humans. In addition to these animals’ small size – about half that of normal “wild type” mice – the scientists quickly noticed that teeny mice produce a lot of urine, often a sign of diabetes.
Beutler is a regental professor and director of the Center for the Genetics of Host Defense. Zhang is an assistant professor of internal medicine who also has an appointment in the center.
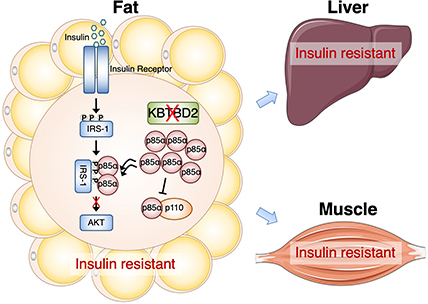
Sure enough, tests showed that these animals had extremely high blood sugar, severe insulin resistance, and high insulin levels that peaked at 8 weeks of age and then gradually declined. They also had abnormally low amounts of body fat but had fatty livers. Transplanting teeny mice with fat tissue from normal mice largely resolved these problems, a sign that KBTBD2 in fat tissue in particular is key to each of them. However, say Beutler and Zhang, it was unclear whether these problems were also rooted in KBTBD2 activity in other insulin-responsive tissues, such as muscle and liver.

To answer this question, the researchers created different mouse mutants in which KBTBD2 was selectively inactivated in the animals’ fat, muscle, or liver. Although each of these rodents grew to a normal size – suggesting that this gene acts through other pathways to regulate body growth – only those with KBTBD2 inactivated in fat cells had some other hallmark characteristics of teeny. These animals had extremely high insulin resistance, although only moderately high blood sugar levels. Although their blood insulin levels were also high, they didn’t decline after 8 weeks of age as in teeny mice.
In addition to having abnormally low body fat like their teeny counterparts, those animals missing KBTBD2 in just their fat cells also had fatty livers, suggesting communication between fat and liver tissue.
Together, say Beutler and Zhang, the findings confirm that KBTBD2 plays a key role in regulating insulin sensitivity and a variety of other activities through its role in fat. However, they also raise important questions about what this gene does elsewhere in the body. KBTBD2 produces a protein that slices up another protein known as p85a, part of a larger protein complex that encourages insulin-sensitive cells to produce sugar transporters on their surfaces. Although it clearly performs this job when produced in fat cells, it doesn’t seem to do this in other parts of the body even though it’s widely expressed in other cell types. It is also unclear what part KBTBD2 plays in keeping teeny mice so small. The researchers plan to explore these questions in future studies.
They also plan to investigate the mechanisms behind why these animals have such extreme insulin resistance, which could have implications for Type 2 diabetes in humans, a disease marked by this characteristic.
“Although we know that insulin resistance is very rarely caused by mutations in the insulin receptor or genes responsible for making other proteins known to participate in glucose uptake, most of it is not understood,” says Beutler, a Nobel Laureate. “Getting a better grasp on the function of KBTBD2 could open a completely new window into how insulin sensitivity is regulated.”
Beutler, who developed a technology for instantly identifying induced germline mutations that cause phenotypes in mice, received the Nobel Prize in Physiology or Medicine in 2011 for his discovery of an important family of receptors that allow mammals to sense infections when they occur, triggering a powerful inflammatory response. He holds the Raymond and Ellen Willie Distinguished Chair in Cancer Research, in Honor of Laverne and Raymond Willie, Sr.
Other UTSW researchers who participated in this study include Thomas Gallagher and Philipp E. Scherer.
This work was supported by grants from the National Institutes of Health (K99 DK115766, R01 AI125581, and U19 AI100627) and the Lyda Hill Foundation.
About UT Southwestern Medical Center
UT Southwestern, one of the premier academic medical centers in the nation, integrates pioneering biomedical research with exceptional clinical care and education. The institution’s faculty has received six Nobel Prizes, and includes 24 members of the National Academy of Sciences, 16 members of the National Academy of Medicine, and 13 Howard Hughes Medical Institute Investigators. The full-time faculty of more than 2,500 is responsible for groundbreaking medical advances and is committed to translating science-driven research quickly to new clinical treatments. UT Southwestern physicians provide care in about 80 specialties to more than 105,000 hospitalized patients, nearly 370,000 emergency room cases, and oversee approximately 3 million outpatient visits a year.
