Finding the Achilles' heel of a killer parasite
Two studies in Science examine the basic biology of schistosomes to uncover vulnerabilities that could lead to new treatments
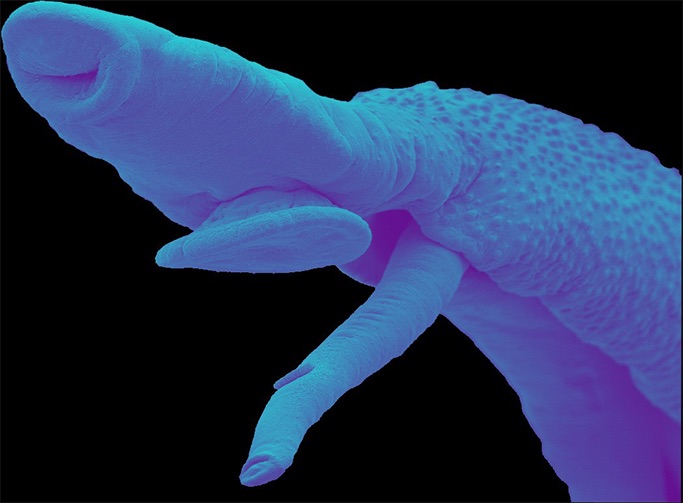
Credit: Dr. James Collins and Ana Vieira
DALLAS – Sept. 24, 2020 – Two studies led by UT Southwestern researchers shed light on the biology and potential vulnerabilities of schistosomes – parasitic flatworms that cause the little-known tropical disease schistosomiasis. The findings, published online today in Science, could change the course of this disease that kills up to 250,000 people a year.

About 240 million people around the world have schistosomiasis – mostly children in Africa, Asia, and South America in populations that represent “the poorest of the poor,” says study leader James J. Collins III, Ph.D., associate professor in UTSW’s department of pharmacology.
Most of those infected survive, but those who die often suffer organ failure or parasite-induced cancer. Symptoms can be serious enough to keep people from living productive lives, Collins says.
The parasite that causes this disease has a complicated life cycle that involves stages in both freshwater snails and mammals. Dwelling in mammalian hosts’ circulatory systems, schistosomes feed on blood and lay copious numbers of eggs, all while causing an array of symptoms including abdominal pain, diarrhea, bloody stool, or blood in the urine. Larval worms are released from snails into water, where the flatworms then may infect humans by penetrating the skin. Schistosomiasis may become a chronic disease that affects the person for years.
Only one drug, praziquantel, is available to treat this condition. However, Collins explains, it is of limited use – it doesn’t kill all intramammalian stages of the schistosome life cycle, and it has a variable cure rate in some endemic settings. There’s been little interest by pharmaceutical companies in developing new drugs for this disease, he adds, because there is no monetary incentive to do so. Consequently, relatively few studies have been devoted to understanding schistosomes’ basic biology, which might reveal inherent weaknesses that could serve as targets for new drugs.
To that end, Collins and his colleagues embarked upon two separate studies – one at the cellular level and another at the molecular level – to better understand these organisms.
In the first study, the researchers delved into the cell types that make up these flatworms. Although the worms are multicellular organisms composed of a variety of unique tissue types, researchers knew little about the different cell populations in these parasites.

With a goal to create an atlas of cell types in Schistosoma mansoni – one of the schistosome species that commonly causes schistosomiasis – Collins and his team used a technique called single-cell RNA sequencing that distinguishes individual cell types based on their unique gene expression patterns. With this method, they identified 68 molecularly unique clusters of cells, including a population of stem cells that form the gut. When the researchers used a targeted approach called RNA interference (RNAi) to shut down the activation of a key gene in these cells, the resulting worms couldn’t digest red blood cells – a key to their growth and a pivotal part of the pathology they cause.
In the second study, the researchers used RNAi to sort out the function of about 20 percent of S. mansoni’s protein coding genes – 2,216 in total. Previously, only a handful of genes in these organisms had been assessed.
By deactivating the genes one by one, Collins and his colleagues identified more than 250 genes crucial for survival. Using a database of pharmacological compounds, the researchers then searched for drugs that had the potential to act on proteins produced by these genes, identifying several compounds with activity on worms. The team also uncovered two protein kinases – a group of proteins renowned for their ability to be targeted by drugs – that are essential for muscle function. When these kinases were inhibited, the worms became paralyzed and eventually died, suggesting that drugs targeting these proteins could eventually treat people with schistosomiasis. A next step in the research will be to search for inhibitors of these proteins.
Collins notes that these strides in understanding the basic biology of schistosomes could eventually lead to new treatments to save untold lives in places where schistosomiasis is endemic.
“This is a very important disease that most people have never heard of,” he says. “We need to invest and invigorate research on these parasites.”
UTSW researchers who contributed to the first study include George Wendt, Lu Zhao, Rui Chen, and Michael L. Reese. UTSW researchers who contributed to the second study include Jipeng Wang, Carlos Paz, Irina Gradinaru, and Julie N. R. Collins.
The first study was supported by grants from the National Institutes of Health (R01 R01AI121037, R01 R01AI150715, R21 R21AI133393, and F30 1F30AI131509-01A1, the Welch Foundation (I-1948-20180324 and I-1936-20170325), the National Science Foundation (MCB1553334), the Burroughs Wellcome Fund, the Wellcome Trust (107475/Z/15/Z), and the Bill and Melinda Gates Foundation (OPP1171488).
The second study was supported by grants from the National Institutes of Health (R01AI121037), the Welch Foundation (I-1948-20180324), the Burroughs Wellcome Fund, and the Wellcome Trust (107475/Z/15/Z and 206194).
James Collins is Rita C. and William P. Clements, Jr. Scholar in Biomedical Research.
About UT Southwestern Medical Center
UT Southwestern, one of the nation’s premier academic medical centers, integrates pioneering biomedical research with exceptional clinical care and education. The institution’s faculty members have received six Nobel Prizes and include 24 members of the National Academy of Sciences, 25 members of the National Academy of Medicine, and 13 Howard Hughes Medical Institute Investigators. The full-time faculty of more than 3,300 is responsible for groundbreaking medical advances and is committed to translating science-driven research quickly to new clinical treatments. UT Southwestern physicians in more than 80 specialties care for more than 143,000 hospitalized patients, attend to more than 470,000 emergency room cases, and oversee nearly 5.3 million outpatient visits a year.
