Research Labs and Groups
Clinician scientists and basic science scientists have established a strong research foundation within the Department of Otolaryngology – Head and Neck Surgery. Our faculty recognize and embrace research as a fundamental value, leading to the development of innovative diagnostic tools and therapies to improve the care and well-being of our current and future patients. Researchers have highly competitive funding, including grants from the National Institutes of Health.
Adrianna Shembel, Ph.D.
Project 1: Sensory Mechanisms in Primary Muscle Tension Dysphonia
Principal investigator: Adrianna Shembel, Ph.D.
Funding source: NIH
Project overview: Primary muscle tension dysphonia (pMTD) is a functional voice disorder that impacts communication, occupation, and quality of life. Although muscle tension and hyperfunction in the laryngeal system are the prevalent theory underlying patient symptoms of vocal effort, fatigue, and discomfort, previous studies comparing well-vetted physiological laryngeal muscle metrics and self-reported symptoms from our lab suggest pMTD may have more to do with an altered sensory system than aberrant motor patterns. The goal of this project is to elucidate the role of sensory mechanisms in pMTD and their relationship to pMTD symptomology.
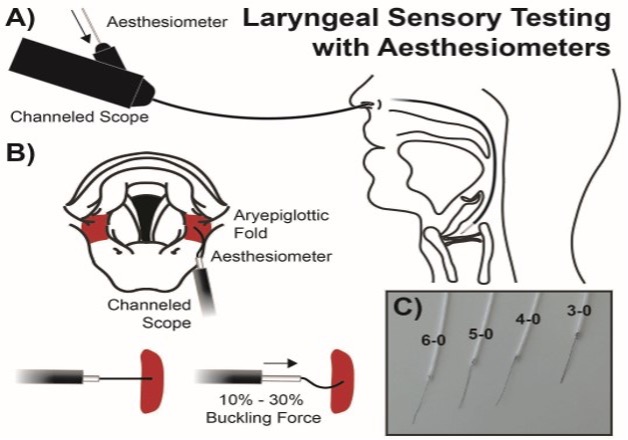
Project 2: Paralaryngeal-Respiratory Spatiotemporal Patterns in Muscle Tension Dysphonia
Principal investigator: Adrianna Shembel, Ph.D.
Funding source: NIH
Project overview: Primary muscle tension dysphonia (pMTD) is a functional voice disorder that negatively affects communication, occupation, and quality of life. Although excessive paralaryngeal muscle activity and altered phonatory breathing patterns are thought to underlie symptoms of vocal effort, fatigue, and discomfort in pMTD, there is conflicting evidence for the role of paralaryngeal-respiratory movement patterns in these clinical presentations due to lack of robust methodological tools to study these relationships. We use novel application of motion capture technology to study these movement patterns in patients with and without pMTD and their relationship to pMTD symptomology.
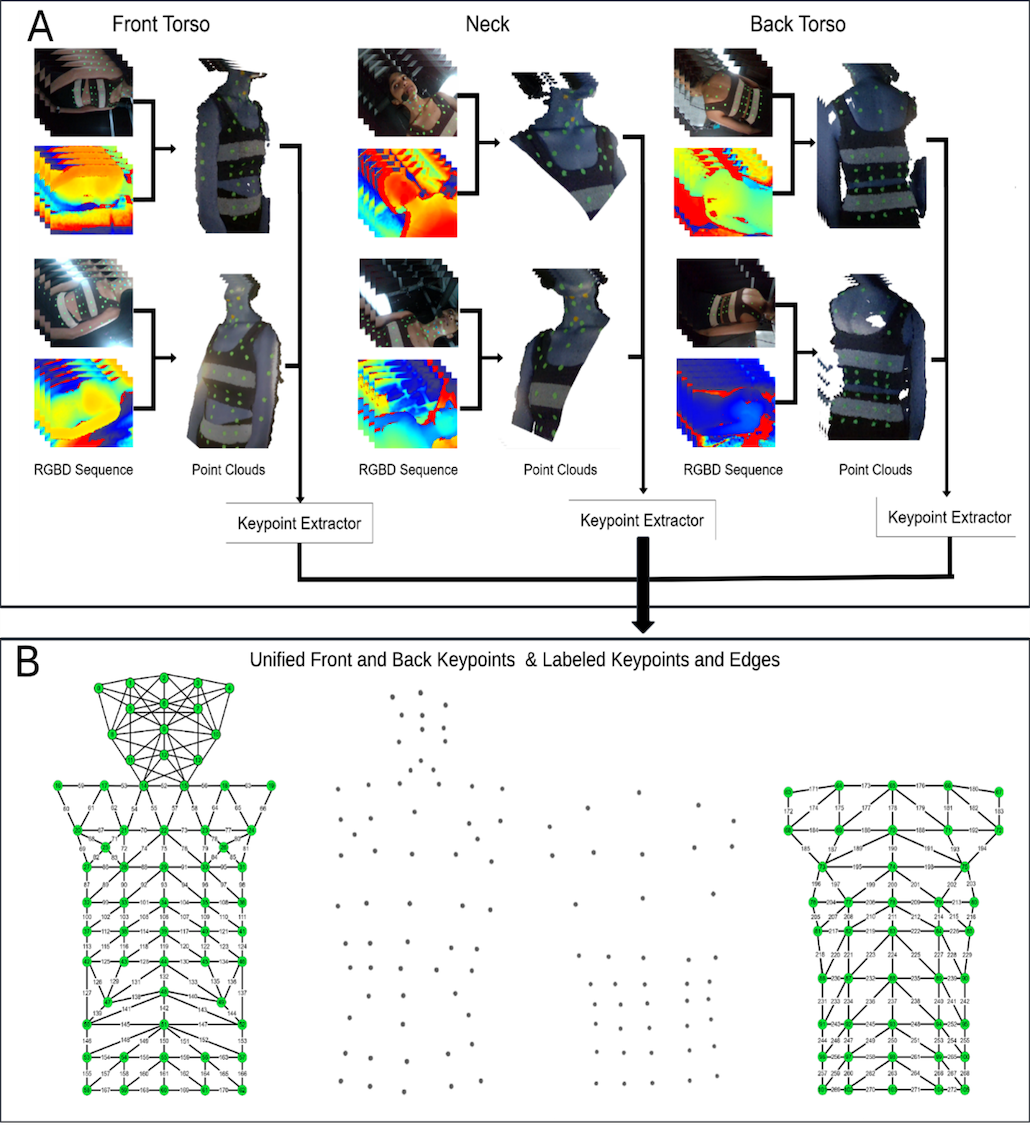
Project 3: Central Modulation–Paired Motor Rehabilitation for Recurrent Laryngeal Nerve Injury
Principal investigator: Adrianna Shembel, Ph.D.
Funding source: NIH
Project overview: The project targets recovery following recurrent laryngeal nerve (RLN) injury, a major cause of vocal fold paralysis. Using a rodent model, this project evaluates whether pairing vagal nerve stimulation (VNS) with motor training can enhance neuroplasticity and improve restoration of vocal fold movement. Positive results could establish a new class of neuromodulation-augmented rehabilitation approaches for RLN injury.
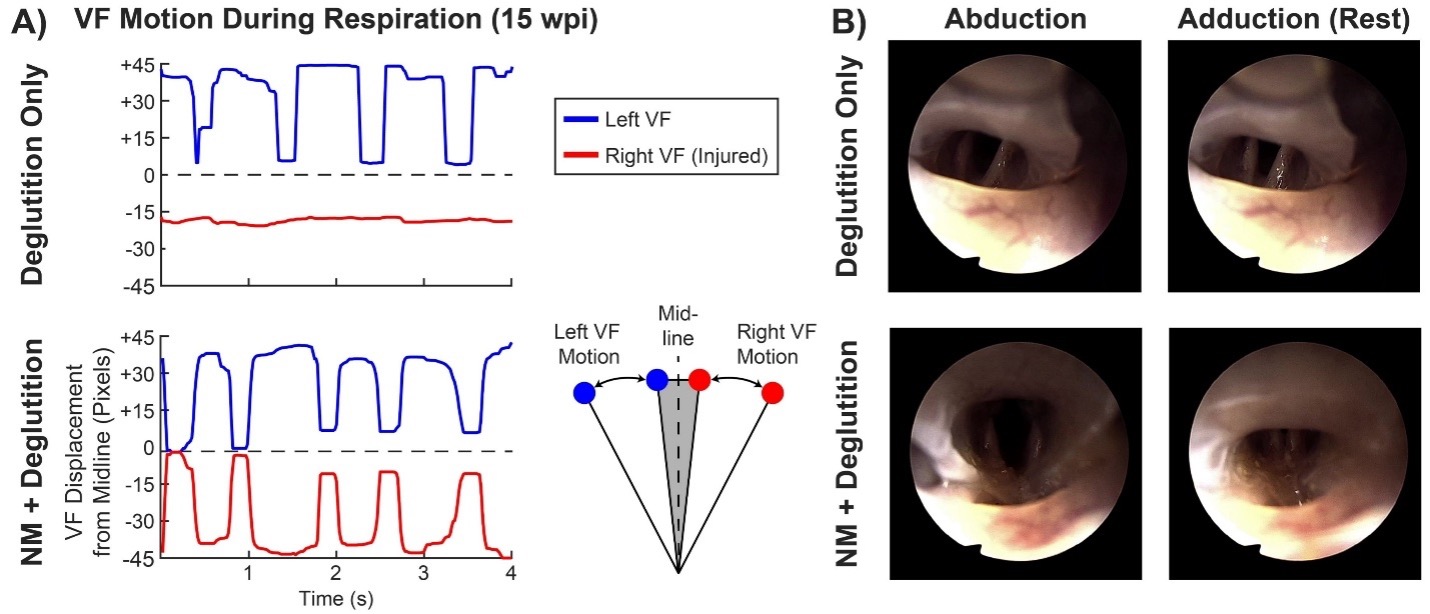
Collectively, Dr. Shembel’s projects provide a multi-system investigation of sensory processing, motor coordination, and neural recovery, positioning the UT Larynx Lab to drive precision-based advances in voice disorder treatment.
Baran Sumer, M.D.
Project: A pH Responsive Transistor-like Nanoprobe for Sensitive Detection of Unknown Primary Cancers of the Head and Neck
Principal investigator: Baran Sumer, M.D.
Funding source: NIH
Project overview: Cancer cells are metabolically active and use the glycolytic pathway to generate energy, resulting in high uptake of glucose and preferential production of lactate (Warburg effect). Exportation of lactic acid/CO2 leads to a pH drop within the tumor microenvironment. Our lab has developed a series of pH-activatable fluorescence probes with different pH transitions attached to different fluorescent dyes, such as rhodamine, Cy5, or ICG. These pH-activatable fluorescence probes give us the ability to image tumors, study tumor acidosis (Warburg effect), and investigate the tumor immune microenvironment (TIME). To investigate potential correlations between TIME and tumor acidosis, our lab used 10X Genomics' Xenium Prime spatial transcriptomics. This has allowed us to analyze differential gene expression in terms of cell type and location at single-cell resolution, as well as comparing HPV+ with HPV- tumors, and unknown primary tumors with their associated lymph node metastasis. Through a novel integration method, our lab has added each sample's associated pH-activatable fluorescence probe data at the single-cell level, providing further avenues for downstream analysis.
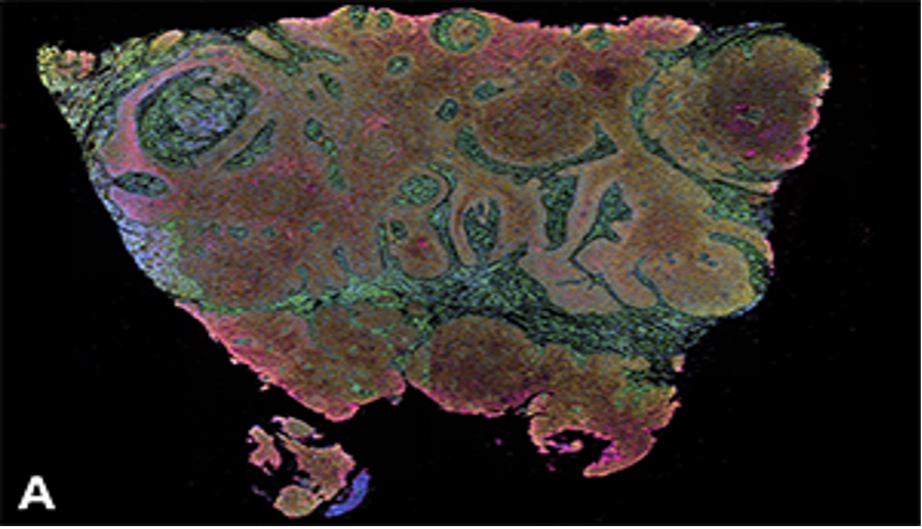
Hitomi Sakano, M.D., Ph.D.
Project: Role of Fragile X Mental Retardation Protein on Gene Expression in Auditory Brainstem Development
Principal investigators: Hitomi Sakano, M.D., Ph.D.
Funding source: NIH
Project overview: Our lab is interested in understanding the molecular mechanisms underlying how our brain adapts or maladapts to auditory stimuli. We are studying these processes in a model for autism (Fragile X syndrome) that exhibits auditory hypersensitivity. We use a variety of techniques, including auditory behavior, immunohistochemistry, and next-generation sequencing. In addition to this, we have collaborations to investigate the features that lead to brain tumor aggressiveness (e.g., glioma) and clinical research related to otologic disorders.
https://labs.utsouthwestern.edu/sakano-lab
Kenneth Lee, M.D., Ph.D.
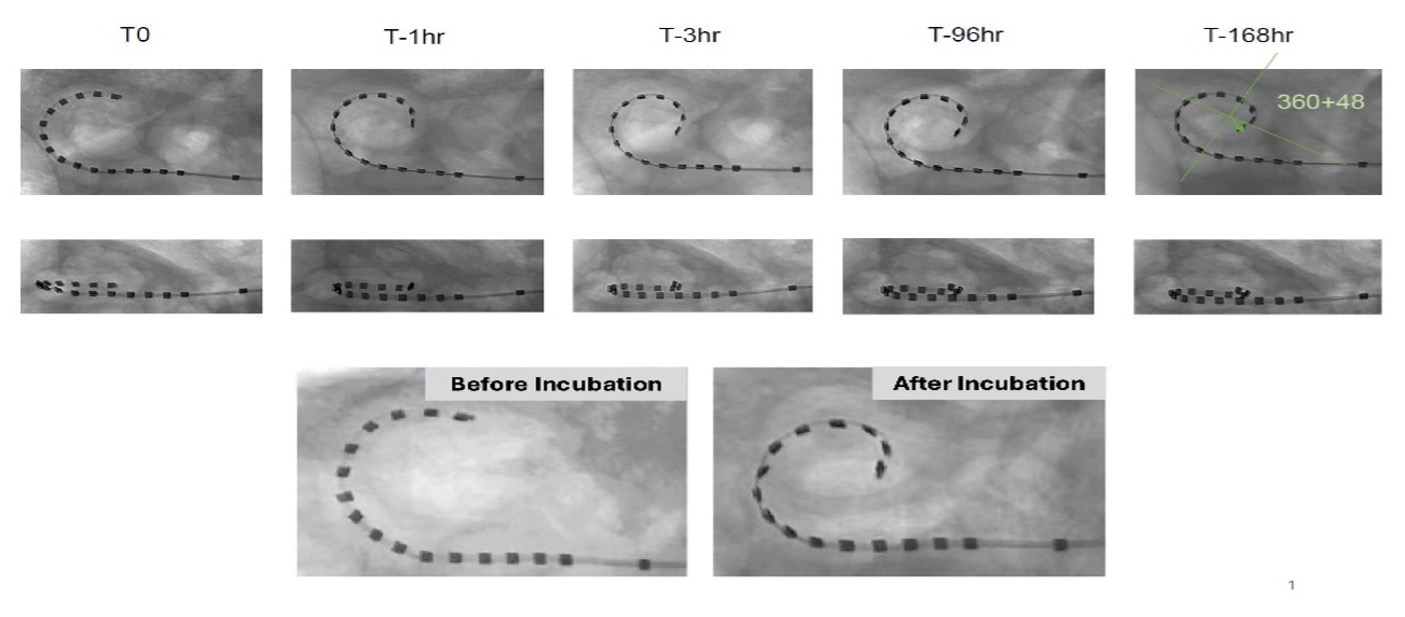
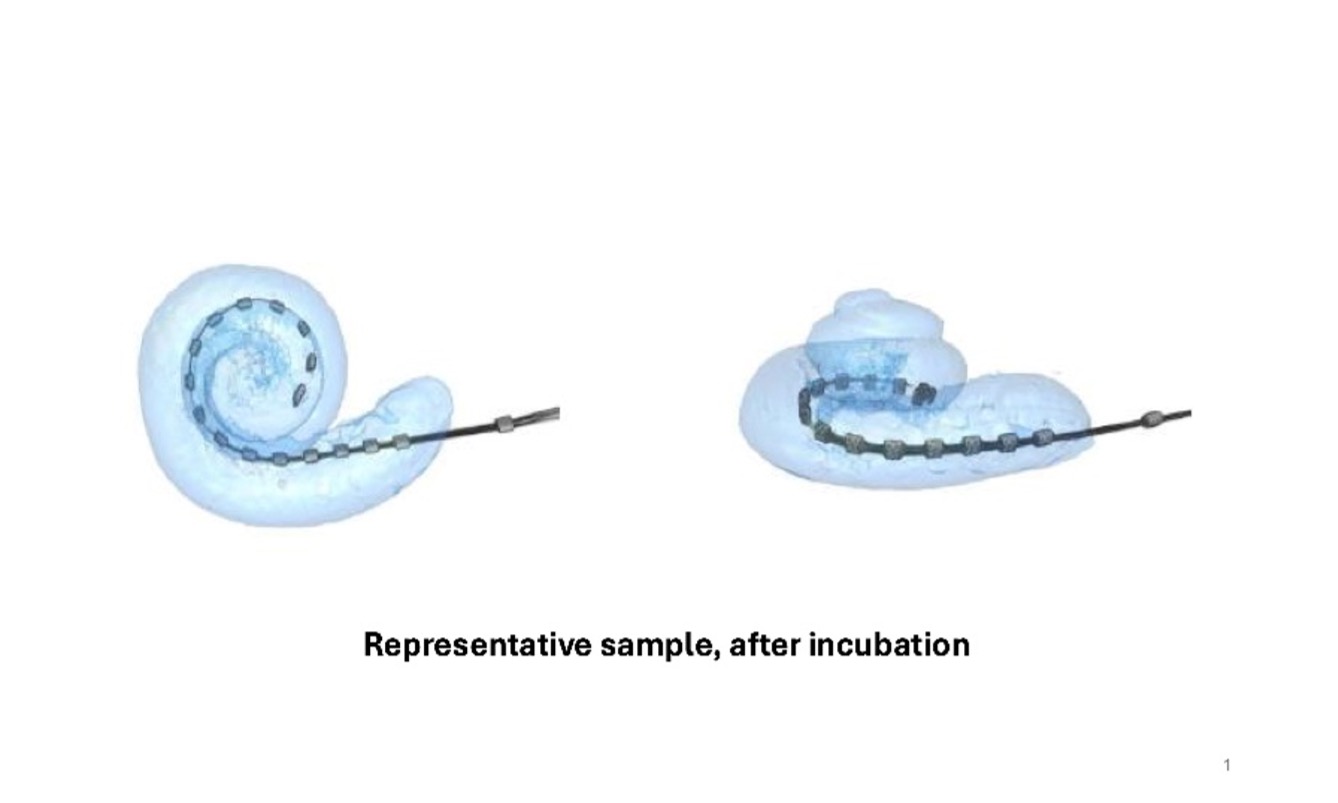
Project: Developing a Novel Self Coiling Cochlear Implant Array with Shape Memory Polymers
Principal investigators: Kenneth Lee, M.D., Ph.D., and Walter Voit
Collaborators: Connie Manz, Jimin Maeng
Funding source: NIH
Project overview: We are developing a novel cochlear implant electrode array using Shape Memory Polymers that is heat sensitive and can change shape based on temperature. Using this polymer, we have created a cochlear implant array that is straight at room temperature but then self-coils to match the turns of the cochlea once inserted and warms to body temperature. With this technology, our novel cochlear implant can be inserted as a straight lateral wall array, but then slowly changes shape and moves medially toward the modiolus. This approach combines the atraumatic advantages of a lateral wall array with the electrophysiological benefits of a perimodiolar array, bringing together the strengths of both types of arrays that are currently available for implantation in patients.
Laura Toles, Ph.D.
Project 1: Longitudinal Investigation of Factors Related to the Development and Rehabilitation of Phonotrauma
Principal investigator: Laura Toles, Ph.D., CCC-SLP
Collaborator: Ted Mau, M.D., Ph.D.
Funding source: NIH
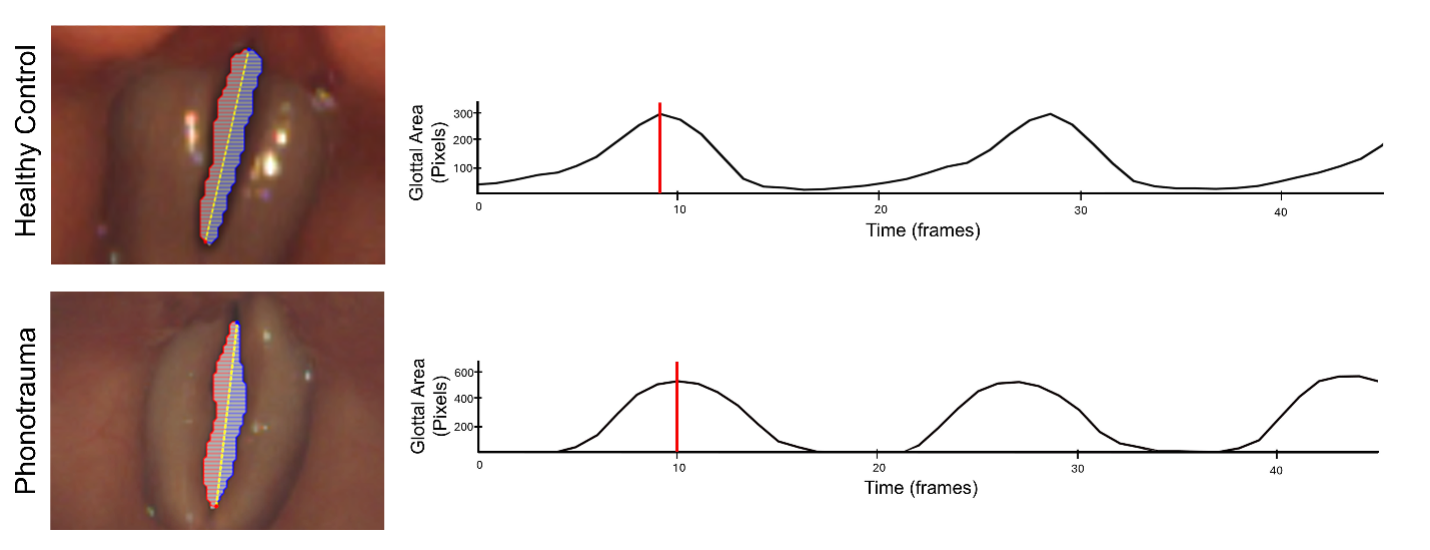
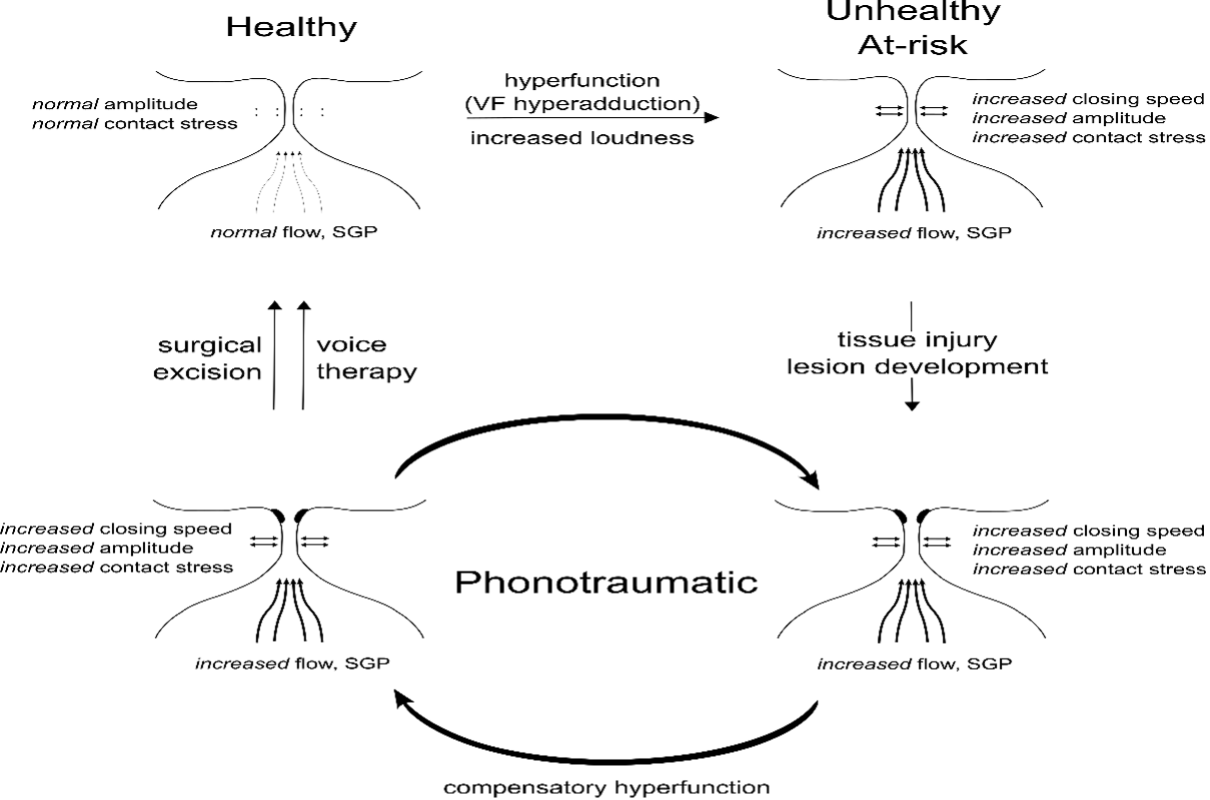
Project overview: As one of the most prevalent behavioral voice disorders, phonotrauma (e.g., vocal fold nodules, polyps, etc.) can have devastating impacts on an individual’s ability to function in their daily life, but it continues to be unclear what factors cause and/or perpetuate these disorders. Thus, it is vital to determine factors related to the development and recovery of phonotrauma in order to prevent and adequately treat these disorders. Using a comprehensive assessment battery that includes high-speed videoendoscopy, ambulatory voice monitoring, and validated psychological trait assessments, the goal of this research is to longitudinally assess the role that psychosocial and behavioral factors play in the development and persistence of phonotrauma in a group of high-risk individuals with the long-term objectives of improving early identification of individuals at risk for phonotrauma and reforming treatment paradigms to improve outcomes.
Project 2: Objective Measures for Clinical Assessment of Voice Disorders
Principal investigator: Laura Toles, Ph.D., CCC-SLP (Site PI)
Collaborator: Cara Stepp, Ph.D. (Overseeing PI at Boston University)
Funding source: NIH
Project overview: Adductor laryngeal dystonia (AdLD) is a neurological voice disorder in which involuntary laryngeal spasms disrupt speech. People with AdLD often experience strained, effortful voice production that negatively affects communication, psychosocial well-being, and quality of life. The current gold-standard treatment - repeated botulinum toxin (botox) injections - provides only temporary relief, and emerging long-lasting therapies cannot be fully evaluated without reliable, sensitive measures of vocal function. However, few clinical outcomes are specific enough to capture the unique symptoms of AdLD, and clinicians frequently struggle to distinguish AdLD from muscle tension dysphonia (MTD), a different voice disorder with overlapping characteristics.
Our research aims to address this gap by developing and validating objective, automated measures that quantify both the primary symptoms of AdLD (laryngeal spasms) and the secondary symptoms (increased laryngeal tension). The current observational study expands this work to a larger cohort. We are examining the physiological validity, sensitivity, reliability, and diagnostic specificity of these measures by comparing individuals with AdLD and both MTD and healthy speakers, evaluating changes before and after treatment. This project will yield robust, automated tools for assessing AdLD, enabling more accurate diagnosis, better tracking of treatment outcomes, and faster translation of new therapies into clinical practice.
Ron Mitchell, M.D.
Project 1: Effects of Hypoglossal Nerve Stimulation on Cognition and Language in Down Syndrome and Obstructive Sleep Apnea
Principal investigator: Ron Mitchell, M.D. (Site PI)
Collaborators: Christopher Hartnick, M.D. (PI); Brian Skotko, M.D. (PI)
Funding source: NIH
Project overview: The goal of the project is to determine the outcomes of hypoglossal nerve stimulation in the treatment of OSA in adolescents with Down Syndrome.
Project 2: ReDIreCTS: Reducing Disparities through Improved Communication in Tonsillectomy and Sleep
Principal investigator: Ron Mitchell, M.D. (Site PI)
Collaborator: Emily Boss, M,D, (PI)
Funding source: NIH
Project overview: The goal of the project is to determine the barriers to improved communications in an ethnically diverse population of children undergoing adenotonsillectomy for the treatment of childhood SDB.
Project 3: Interaction of Mycobacterium tuberculosis with upper airway mucosal cells
Collaborator: Ron Mitchell, M.D.
Principal investigator: Michael Shiloh, M.D., Ph.D. (PI)
Funding source: NIH
Project overview: The airway mucosa is the initial site of interaction with Mycobacterium tuberculosis. While the mucosa itself forms a critical barrier to infection, it is also the anatomic location where cells of the mucosal, innate and adaptive immune systems converge to orchestrate protective immediate and long-term responses. We have demonstrated that M. tuberculosis can migrate and enter the mucosa associated lymphoid tissue via a rare, specialized cell type called a microfold cell. Here we propose to better characterize features of the airway M cell, establish how it communicates with other epithelial and immune cells, determine the mechanism by which the bacteria traverse the cell and the immunologic consequences of early microfold cell contact with M. tuberculosis. This approach is novel because it represents the first broad characterization of the role of airway microfold cells to host defense against M. tuberculosis.
Project 4: A mechanistic understanding of treatment-related outcomes of sleep disordered breathing using functional near infrared spectroscopy
Principal investigator: Ron Mitchell, M.D. (Site PI)
Collaborator: Amal Isaiah, M.D., Ph.D. (PI)
Funding source: NIH
Project overview: The goal of the project is to use functional near infrared spectroscopy and leading-edge analytic tools to understand the outcomes of treatment of sleep-disordered breathing from a neurobiologic standpoint.
Project 5: Treatment of Obstructive Sleep Apnea with Personalized Surgery in Children with Down Syndrome (TOPS-DS)
Principal investigator: Ron Mitchell, M.D. (Site PI)
Collaborator: Derek Lam, M.D. (PI)
Funding source: NIH
Project overview: The overall objective of this randomized clinical trial is to test the effectiveness of a novel personalized approach to the surgical treatment of OSA in children with Down Syndrome.
