Study identifies how certain drugs alter the metabolism of pancreatic cancer cells, revealing a new therapeutic target
DALLAS – Jan. 21, 2016 – UT Southwestern Medical Center researchers have found that cancer drugs known as CDK4/6-inhibitors alter the metabolism of pancreatic cancer cells, revealing a biologic vulnerability that could be exploited for therapeutic gain. The findings were published today in Cell Reports.

Because pancreatic cancer has one of the worst prognoses of any cancer and is the third leading cause of cancer deaths in the U.S., according to the National Cancer Institute, researchers for years have sought to find better treatment options.
Last year, the FDA approved the first cyclin-dependent kinase 4/6 (CDK4/6) inhibitor for treating a certain type of advanced breast cancer. This class of drugs has been widely studied in clinical trials for many other types of cancer, including pancreatic cancer. CDK 4/6 inhibitors are cytostatic, meaning they work by preventing cancer cells from growing and dividing.
“On the one hand, that’s great, because the tumor won’t grow, but on the other hand, the patient still has a tumor, which will eventually become resistant to those drugs,” said study senior author Dr. Erik Knudsen, Professor of Internal Medicine in the Eugene McDermott Center for Human Growth and Development at UT Southwestern.
“There’s a lot of interest in better understanding the biology behind CDK4/6 inhibitors – and in finding out whether we can use that information to kill tumors instead of simply stopping their growth,” added Dr. Agnieszka Witkiewicz, also in the McDermott Center and an Associate Professor of Pathology.
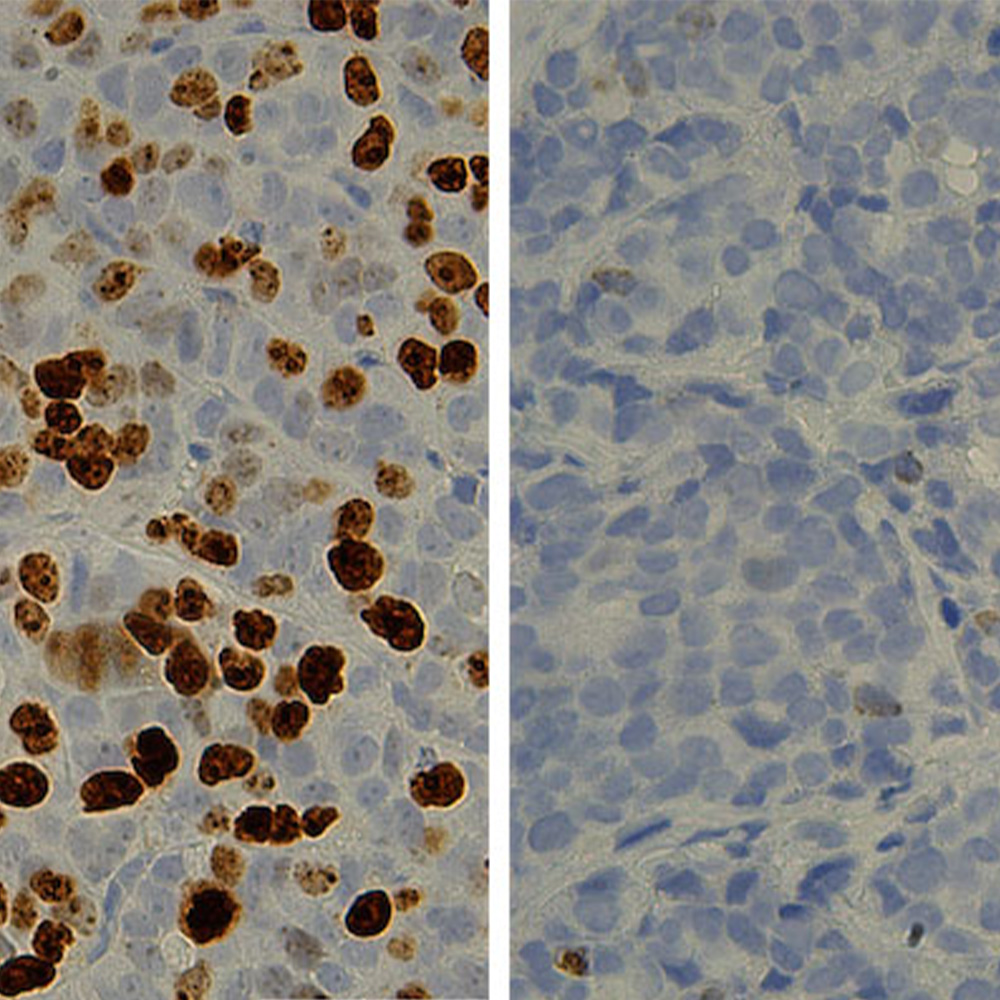
In this study, the research team treated human pancreatic cancer cells and tumors grown in mice with CDK4/6-inhibiting drugs. Surprisingly, they found that when tumor cells were treated with CDK4/6 inhibitors, the cells’ metabolism – the way cancer tumors get energy – became more active.
“Now we can try attacking specific aspects of CDK4/6-induced metabolic programming,” said Dr. Knudsen, also a member of UT Southwestern’s Harold C. Simmons Comprehensive Cancer Center, along with Dr. Witkiewicz. “For example, by targeting altered tumor metabolism, we could potentially turn the cytostatic effect of CDK4/6 inhibitors into a cytotoxic effect that actually kills the cancer cells.”
The upshot is that by disrupting a tumor’s cell cycle with CDK4/6 inhibitors and then targeting the altered metabolism with other drugs – such as mTOR inhibitors – it may be possible to positively impact cancer treatment.
“These data yield valuable new insights into the cross talk between CDK inhibitors, signaling pathways, and tumor metabolism in pancreatic cancer, opening up some interesting new possibilities for treatment that could be evaluated in clinical trials,” Dr. Witkiewicz said. “The real goal is that this work – as well as ongoing studies – will benefit patients with pancreatic cancer.”
Other researchers from the McDermott Center involved in this study were graduate student Jorge Franco and computational biologist Uthra Balaji. Researchers from Massachusetts Institute of Technology’s Whitehead Institute also contributed.
The study was supported by grants from the National Institutes of Health.
About UT Southwestern Medical Center
UT Southwestern, one of the premier academic medical centers in the nation, integrates pioneering biomedical research with exceptional clinical care and education. The institution’s faculty has included six who have been awarded Nobel Prizes since 1985. The faculty of almost 2,800 is responsible for groundbreaking medical advances and is committed to translating science-driven research quickly to new clinical treatments. UT Southwestern physicians provide medical care in about 80 specialties to more than 92,000 hospitalized patients and oversee approximately 2.2 million outpatient visits a year.
###
Media Contact: Debbie Bolles
214-648-3404
debbie.bolles@utsouthwestern.edu
To automatically receive news releases from UT Southwestern via email,
subscribe at www.utsouthwestern.edu/receivenews