Researchers lead collaborative charge to uncover genetic diversity of pancreatic cancer

DALLAS – April 9, 2015 –A genetic analysis led by UT Southwestern Medical Center researchers suggests that most pancreatic cancers harbor genetic alterations that could be targeted by existing drugs, using their genetic features as a roadmap for treatment. The findings support a precision approach to treating pancreatic cancer, the fourth most deadly cancer for both men and women.
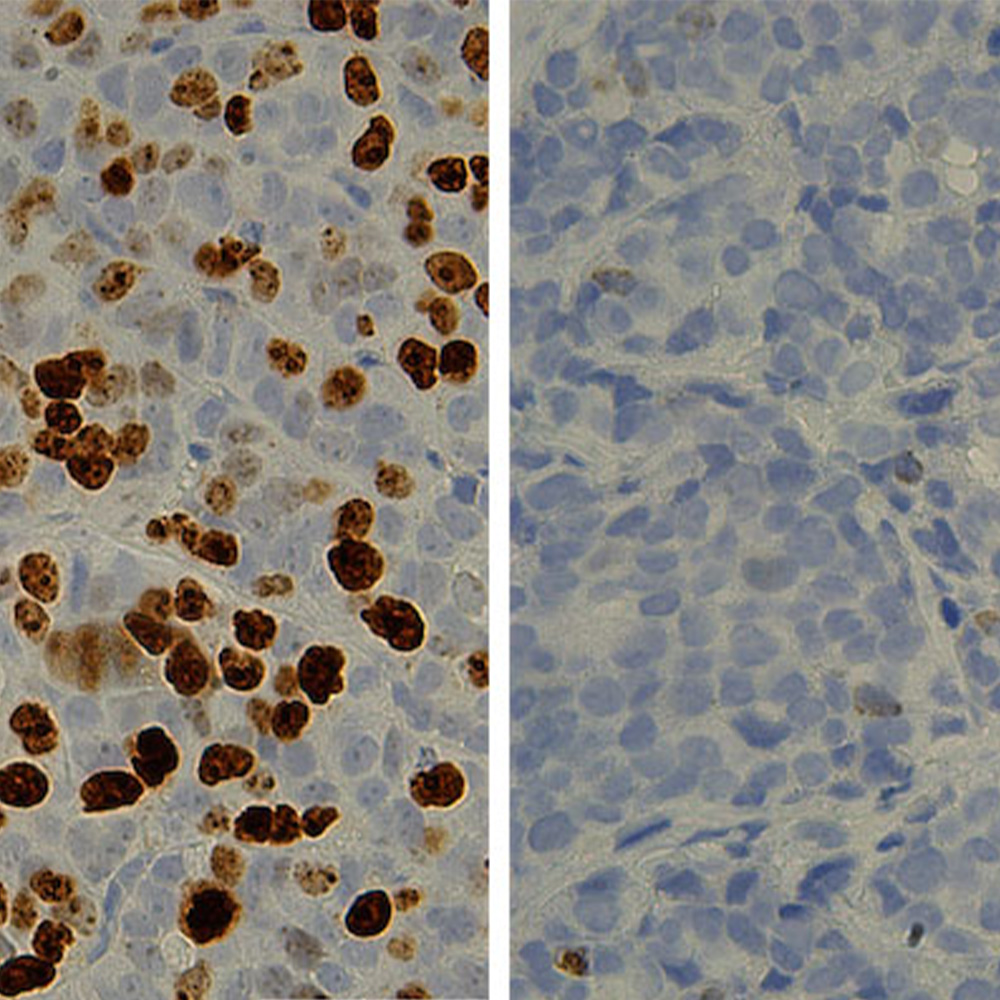
A comprehensive DNA sequencing of pancreatic cancer cases revealed not only a plethora of damaged genes, but potential diagnostic biomarkers that could help identify those with longer or shorter survival, and provide opportunity for new therapeutic interventions. The new findings are published in Nature Communications.
“We identified a wealth of genetic diversity, including multiple mutated genes that were previously unknown to pancreatic cancer − an important step in gaining a better understanding of this difficult and particularly deadly disease,” said lead author Dr. Agnieszka Witkiewicz, Associate Professor of Pathology and a member of the Harold C. Simmons Comprehensive Cancer Center at UT Southwestern. “Importantly, the team was able to identify several genes that may be able to help us to predict outcomes in certain circumstances or serve as good candidates for therapeutic efforts.”
Researchers have long hoped that genetic analysis would provide insight into the biology of pancreatic cancer and define new targets for more effective treatment. Achieving this goal has been hampered by the technical difficulty of isolating pure cancer cells out of the tumor tissue that contains both tumor cells as well as normal cells. The new study overcame this limitation by selectively dissecting cancer cells from pieces of tumor tissue. This method was applied to specifically determine the genetic features of 109 different tumors.
The data showed that the genetic architecture of pancreatic cancer is complex, and each patient’s tumor was found to be unique. The genetic features illuminated ways in which the disease arises, defined events associated with survival, and yielded potential targets for therapeutic intervention.
“While we suspect that genetics can be used as the basis of targeted treatments, this point will only be proven through extensive research and clinical studies, hopefully leading to improved outcomes for patients,” said senior author Dr. Erik Knudsen, Professor of Pathology, and member of the Simmons Cancer Center who holds the Dr. Charles T. Ashworth Professorship in Pathology. “I am considerably more optimistic of the utility of a genetically targeted therapy for pancreatic cancer today than when we began this work.”
Pancreatic cancer is particularly difficult to treat, and is often diagnosed at a late stage when it is no longer amenable to surgical removal. Chemotherapy has a modest effect, and unfortunately the disease progresses in the vast majority of cases. Therefore, new therapeutic regimens are urgently needed.
“Pancreatic cancer will surpass breast and prostate to become the third-leading cause of cancer-related deaths in the next 15 years. If we want to change the death rate, we need to increase the investment in understanding the biology of pancreatic cancer and identifying novel treatment strategies,” said co-author and pancreatic cancer surgeon Dr. Michael Choti, Chairman of Surgery, who holds the Hall and Mary Lucile Shannon Distinguished Chair in Surgery.
The study identified a multitude of events that could be selectively targeted in the context of individual pancreatic tumors. For example, the study defined cases of pancreatic cancer that are driven by a gene called BRAF, for which there are FDA-approved drugs, and showed that such drugs were selectively effective against BRAF-mutated pancreatic cancer cells. Similarly, they defined deficits in other features of the tumor cell that can be treated by select drugs.
While these are exciting discoveries for a disease with such a poor prognosis, Dr. Witkiewicz voiced caution.
“Most pancreatic cancers exhibit multiple genetic alterations; therefore, it is likely that combination approaches targeting multiple pathways will be required for effective disease control,” she said.
Additional UT Southwestern researchers involved included Dr. Prasad Koduru, Professor of Pathology and Director of the Genetics Diagnostics Laboratories; Dr. Michael A. White, Professor of Cell Biology, who holds the Grant A. Dove Distinguished Chair for Research in Oncology and the Sherry Wigley Crow Cancer Research Endowed Chair in Honor of Robert Lewis Kirby, M.D.; Dr. John Mansour, Associate Professor of Surgery; Dr. Adam Yopp, Assistant Professor of Surgery; Uthra Balaji, computational biologist; Dr. GuemHee Baek, postdoctoral researcher; and Elizabeth McMillan, student research assistant in Cell Biology.
Researchers from the Department of Pathology and Surgery from Thomas Jefferson University in Philadelphia, Pa., and the Eppley Institute for Research in Cancer and Allied Diseases at the University of Nebraska Medical Center in Omaha also contributed to the research.
The research was supported by grants from the National Cancer Institute and the Cancer Prevention and Research Institute of Texas. Support for tissue acquisition and processing was through the Harold C. Simmons Comprehensive Cancer Center Tissue Management Resource.
UT Southwestern’s Harold C. Simmons Comprehensive Cancer Center is the only National Cancer Institute-designated cancer center in North Texas and one of just 68 NCI-designated cancer centers in the nation. The Simmons Cancer Center includes 13 major cancer care programs with a focus on treating the whole individual with innovative treatments, while fostering groundbreaking research that has the potential to improve patient care and prevention of cancer worldwide. In addition, the Center’s education and training programs support and develop the next generation of cancer researchers and clinicians.
The Simmons Cancer Center is among only 30 U.S. cancer research centers to be named a National Clinical Trials Network Lead Academic Participating Site, a prestigious new designation by the NCI, and the only Cancer Center in North Texas to be so designated. The designation and associated funding is designed to bolster the cancer center’s clinical cancer research for adults and to provide patients access to cancer research trials sponsored by the NCI, where promising new drugs often are tested.
About UT Southwestern Medical Center
UT Southwestern, one of the premier academic medical centers in the nation, integrates pioneering biomedical research with exceptional clinical care and education. The institution’s faculty includes many distinguished members, including six who have been awarded Nobel Prizes since 1985. Numbering approximately 2,800, the faculty is responsible for groundbreaking medical advances and is committed to translating science-driven research quickly to new clinical treatments. UT Southwestern physicians provide medical care in 40 specialties to about 92,000 hospitalized patients and oversee approximately 2.1 million outpatient visits a year.
###
Media Contact: Lori Sundeen Soderbergh
214-648-3404
lori.soderbergh@utsouthwestern.edu
To automatically receive news releases from UT Southwestern via email, subscribe at www.utsouthwestern.edu/receivenews