Study reveals endocrine-producing microenvironment in embryonic pancreas

DALLAS – Jan. 22, 2018 – UT Southwestern researchers report that development of the endocrine pancreas is promoted by a weblike network of epithelial tubes that exist only transiently in the developing embryo. The findings, published online by Genes & Development, suggest that a deeper understanding of this endocrine differentiation “niche” could propel development of novel treatments for diabetes.
Researchers from the Departments of Molecular Biology and Internal Medicine (Division of Nephrology) collaborated on the investigation, which was co-led by Drs. Ondine Cleaver and Denise K. Marciano. Below, study first author and graduate student D. Berfin Azizoglu, Dr. Cleaver, an Associate Professor of Molecular Biology and a member of the Hamon Center for Regenerative Science and Medicine, Dr. Marciano, Assistant Professor of Internal Medicine, and postdoctoral researcher Dr. Caitlin Braitsch discuss the findings of their study.
Q: What advance did the team make?
Our group showed that the generation of pancreatic endocrine cells, including the beta cells responsible for producing insulin in our bodies, is promoted by a specialized microenvironment within the embryonic pancreas. Altering this microenvironment can alter endocrine mass.
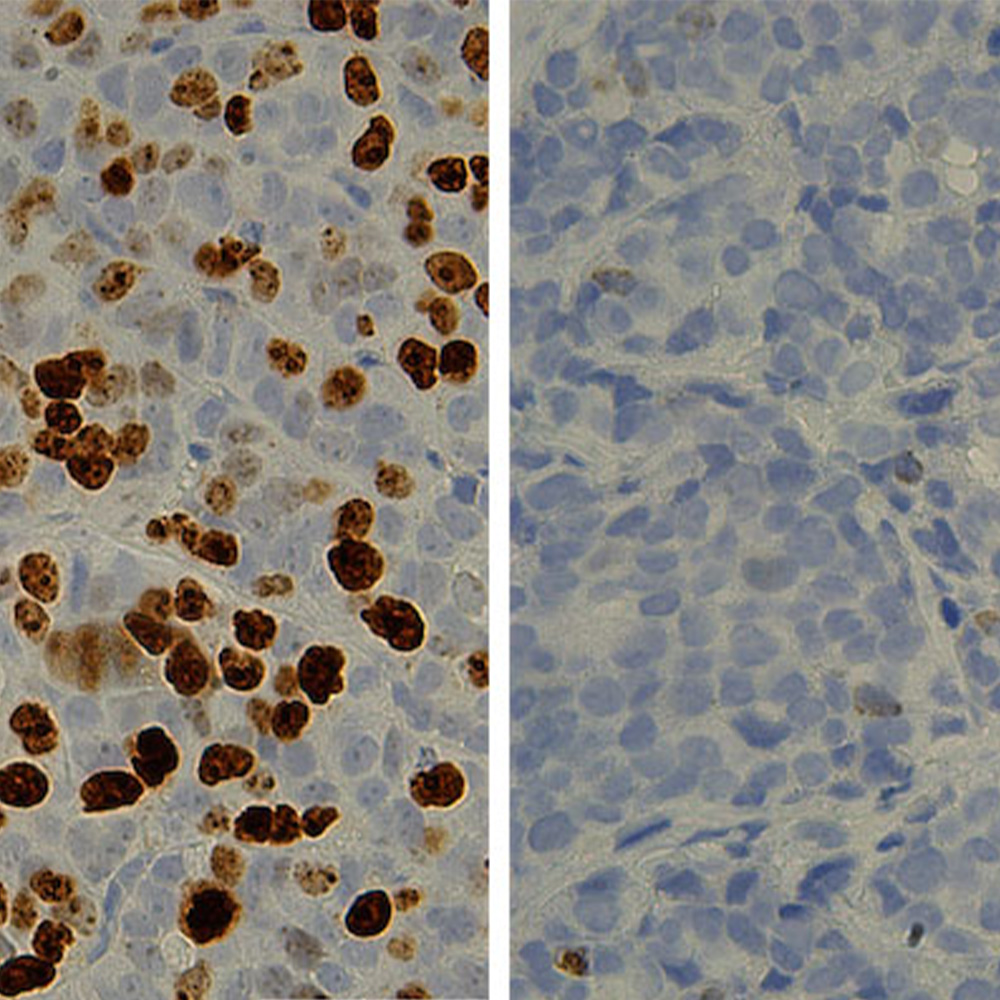
Endocrine cells exist in clusters called “islets of Langerhans.” It is these cells that are destroyed or become nonfunctional in diabetic patients. We discovered that these cells depend on the architecture of their microenvironment and that the weblike network of pancreatic tubes serves as a functional microenvironment for the production of endocrine cells. Previous research has pinpointed this network as the location where an endocrine progenitor cell (stem cell) arises during development.
We altered this microenvironment by deleting Afadin, which is a protein required for tube remodeling. This alteration revealed that increasing the overall volume of these tubes during pancreatic development resulted in an increased mass of endocrine cells, suggesting that these tubes act as a promoter of endocrine cell development.
Q: Why is it an important or key step forward for researchers?
Pancreatic endocrine cell development has been a major research subject for many years and it has informed studies aimed at generating replacement beta cells, which are the cells that become nonfunctional in diabetes. Many key steps of endocrine cell differentiation have been well-characterized, but whether the architecture of the microenvironment containing the progenitors is important for proper endocrine development has remained in question. Our work shows that developing pancreatic tubes organize into a weblike network (as opposed to a branching tree of tubes found in the mature pancreas) and create a home that promotes endocrine cell generation. Formation of more of these tubes results in more endocrine cells.
Q: What are the research implications for diseases or conditions, or for providing a new avenue of investigation?
Endocrine cell deficiency leads to diabetes, which can theoretically be cured by providing functional endocrine cells to patients. Recent advances in stem cell biology have enabled us to make endocrine-like cells in the lab. However, these cells have thus far failed to do the job and need further improvement to become therapeutically viable.
Our work uncovers important characteristics of the birthplace of endocrine cells, and shows that enlargement of this microenvironment induces generation of more endocrine cells. These findings suggest that lab-made endocrine cells could be improved by developing these cells in a microenvironment that resembles their birthplace. This suggestion is supported by a recent boost in endocrine cell generation when the cells were grown in a 3-dimensional matrix, demonstrating that tissue conformation is influential to proper endocrine expansion and differentiation.
Q: How will this research potentially help patients in the future?
By recreating the microenvironment for endocrine cell development in the lab, we may be able to achieve a long-standing goal in diabetes research – to efficiently produce fully functional glucose-responsive endocrine cells from a patient’s stem cells. These cells then could be transplanted back into the patient, and there produce insulin normally.
Q: How does this study help other scientists?
For decades, researchers in the pancreas development and diabetes fields have been trying to understand how endocrine cells are made. Pancreatic endocrine cells were known to emerge along pancreatic tubes, but since these tubes are spread throughout the organ, the exact location of endocrine cell emergence was difficult to pinpoint. We now have evidence that endocrine development is promoted specifically by a set of interconnected, weblike tubes which exist only transiently in the embryonic pancreas. By studying these tubes, their microenvironment, and their cellular and molecular components, we hope to learn about the conditions necessary to produce fully functional endocrine cells.
Q: What cautions if any should be considered?
To understand whether the weblike tubes promote endocrine development, we have used a genetic approach that arrested tubes in a weblike conformation, whereas normally, they are in this conformation for only a short period of time. This perturbation had other effects, such as enlargement of tube diameter, tube discontinuity, and reduction in organ size. These aspects did not seem to hamper endocrine cell generation; however, we cannot rule out the possibility that these alterations contribute to the increased endocrine mass through unknown mechanisms. Further studies that employ different approaches to disturb the tube microenvironment and other architectural aspects of the pancreatic progenitor epithelium will be needed to determine their effects on endocrine cell development.
About UT Southwestern Medical Center
UT Southwestern, one of the premier academic medical centers in the nation, integrates pioneering biomedical research with exceptional clinical care and education. The institution’s faculty has received six Nobel Prizes, and includes 22 members of the National Academy of Sciences, 18 members of the National Academy of Medicine, and 14 Howard Hughes Medical Institute Investigators. The faculty of more than 2,700 is responsible for groundbreaking medical advances and is committed to translating science-driven research quickly to new clinical treatments. UT Southwestern physicians provide care in about 80 specialties to more than 100,000 hospitalized patients, 600,000 emergency room cases, and oversee approximately 2.2 million outpatient visits a year.