DNA vaccine reduces both toxic proteins linked to Alzheimer’s

A study by UT Southwestern’s Peter O’Donnell Jr. Brain Institute shows that a vaccine delivered to the skin prompts an immune response that reduces buildup of harmful tau and beta-amyloid – without triggering severe brain swelling that earlier antibody treatments caused in some patients.
“This study is the culmination of a decade of research that has repeatedly demonstrated that this vaccine can effectively and safely target in animal models what we think may cause Alzheimer’s disease,” said Dr. Roger Rosenberg, founding Director of the Alzheimer’s Disease Center and Professor of Physiology, and Neurology and Neurotherapeutics. “I believe we’re getting close to testing this therapy in people.”
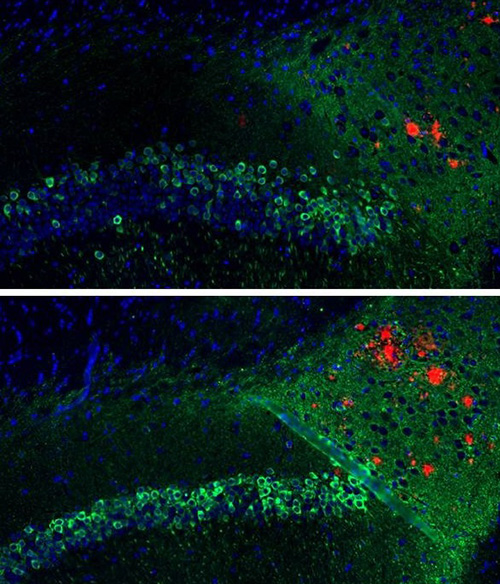
The research published recently in Alzheimer’s Research and Therapy demonstrates how a vaccine containing DNA coding for a segment of beta-amyloid also reduces tau in mice modeled to have Alzheimer’s disease. In addition, the vaccine elicits a different immune response that may be safe for humans. Two previous studies from Dr. Rosenberg’s lab showed similar immune responses in rabbits and monkeys.
The vaccine is on a shortlist of promising antibody treatments aimed at protecting against both types of proteins that kill brain cells as they spread in deadly plaques and tangles on the brains of Alzheimer’s disease patients.
Although earlier research established that antibodies significantly reduce amyloid buildup in the brain, Dr. Rosenberg’s team needed to find a safe way to introduce them into the body. A vaccine developed elsewhere showed promise in the early 2000s, but when tested in humans, it caused brain swelling in some patients.
Dr. Rosenberg’s idea was to start with DNA coding for amyloid and inject it into the skin rather than the muscle to produce a different kind of immune response. The injected skin cells make a three-molecule chain of beta-amyloid (Aβ42), and the body responds by producing antibodies that inhibit the buildup of amyloid and indirectly also of tau.
The latest study – consisting of four cohorts of between 15 and 24 mice each – shows the vaccine prompted a 40 percent reduction in beta-amyloid and up to a 50 percent reduction in tau, with no adverse immune response. Dr. Rosenberg’s team predicts that if amyloid and tau are indeed the cause of Alzheimer’s disease, achieving these reductions in humans could have major therapeutic value.
“If the onset of the disease could be delayed by even five years, that would be enormous for the patients and their families,” said Dr. Doris Lambracht-Washington, the study’s senior author and Assistant Professor of Neurology and Neurotherapeutics.
Dr. Lambracht-Washington presented the preliminary findings on tau reduction in mice in 2016 at the Alzheimer’s Association International Conference and was awarded a grant by the UT Southwestern Circle of Friends to continue the research.
The research was also supported by the National Institutes of Health; the Zale Foundation; the Rudman Foundation; the Presbyterian Village North Foundation; Freiberger, Losinger, and Denker Family Funds; Triumph Over Alzheimer’s; and AWARE.
Dr. Rosenberg holds The Abe (Brunky), Morris and William Zale Distinguished Chair in Neurology.


