C(PRIT) into the future
Three UT Southwestern researchers. Three research strategies. One far-reaching lab bench-to-bedside project to improve anti-cancer immunotherapy.

Backed by a four-year, $6 million grant from the Cancer Prevention and Research Institute of Texas (CPRIT), Dr. Yang-Xin Fu, Dr. Zhijian “James” Chen, and Dr. Raquibul Hannan are taking a unique multipronged approach to study how radiation activates the immune system to fight cancer. They want to learn more about how the cGAS-STING immune response pathway bridges the gap from innate immunity to adaptive, tumor-specific immunity, and they want to figure out how to enhance and harness that response.
The investigation may also provide insights into why some patients develop resistance to conventional anti-cancer therapies and to immunotherapies – leading to potential strategies for overcoming this resistance.
The innate immune system senses initial threats and launches the body’s first response to them. The classical view – that the body’s second immune system, the adaptive one, battles recurring hazards to which the body is already conditioned to respond – raises the question: What is the bridge between the body’s two immune systems?
“This project is absolutely unique. Dr. Chen’s background is in biochemistry and innate immunity. Dr. Fu’s background is in tumor immunology. And I am a physician-scientist.”
A study by Drs. Fu and Chen published in the journal Immunity in August 2017 found that cGAS – the molecular DNA sensor that Dr. Chen identified in his UT Southwestern lab – plays a critical role in that bridge. The study contains the first report that tumors stressed by cancer immunotherapy release their mitochondrial DNA into nearby immune cells, triggering a host alert system via the cGAS-STING pathway. The Hannan laboratory reported in the Proceedings of the National Academy of Sciences in 2017 that one of the early events in radiation-induced anti-tumor immunity is the recruitment to the tumor of neutrophils, the most abundant type of white bloods cells in the body and a hallmark of the innate immune system.
Combining unique skills
“This project is absolutely unique. Dr. Chen’s background is in biochemistry and innate immunity. Dr. Fu’s background is in tumor immunology. And I am a physician-scientist,” says Dr. Hannan, Associate Professor of Radiation Oncology and Immunology with the Harold C. Simmons Comprehensive Cancer Center.
Within the grant the researchers have a pipeline going all the way from the laboratory to the bedside and back, says Dr. Hannan, who studies tissues from patients in his clinical research to establish or confirm mechanisms that have been identified in the lab.
“If there’s a challenge in the clinic, then we can go back to the lab to investigate it. We think having the flexibility to go back and forth will make the outcomes of our three projects much more robust and ready for clinical application,” he adds.
Dr. Chen – Professor of Molecular Biology, Director of the Center for Inflammation Research, and an Investigator in the Howard Hughes Medical Institute – recently won a prestigious 2019 Breakthrough Prize in Life Sciences for his discovery of the cGAS enzyme that launches the body’s immune defense against infections and cancers. That enzyme patrols the cell’s interior and triggers the immune system in response to DNA located in parts of the cellular interior where that genetic material should not exist.
Overcoming resistance
One of the most daunting challenges to effective treatment is the ability of cancer cells to develop resistance to conventional therapies. Strategic combinations of different treatments hold the greatest promise for overcoming therapeutic resistance, Dr. Hannan says.
In addition, although there are now multiple forms of cancer immunotherapy – treatments designed to increase patients’ immune responses – their effects remain inconsistent. When patients respond, the results are often amazing. Yet, only 15-20 percent of patients respond.
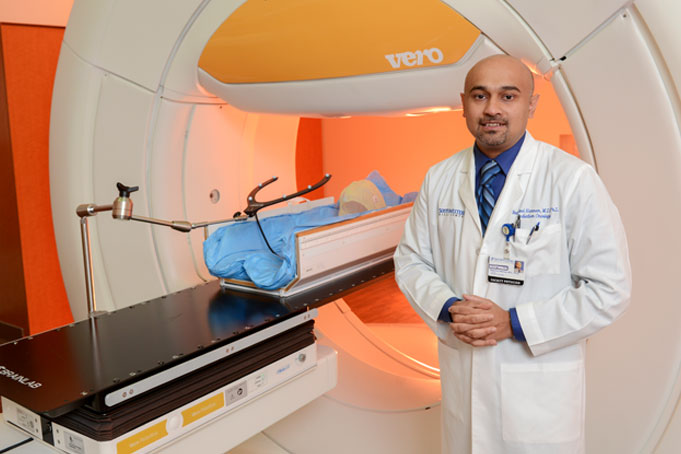
Targeted radiation therapy has been shown both to kill cancer cells by damaging their DNA and to alert the immune system to the presence of cancer cells, he says. “In fact, it appears that highly targeted radiation like the stereotactic radiotherapy we are pioneering in Radiation Oncology at UT Southwestern can act as a ‘cancer vaccine’ to damage cancer cells’ DNA and to fight cells that have metastasized to distant sites in the body.”
However, the mechanisms by which radiation stimulates the immune system are poorly understood. The three studies funded by the CPRIT grant will explore these mechanisms, with the goal of designing more effective therapeutic combinations of immunotherapy and radiation to advance cancer treatment.
A three-pronged inquiry
The grant is divided into three related projects that cover the research spectrum:
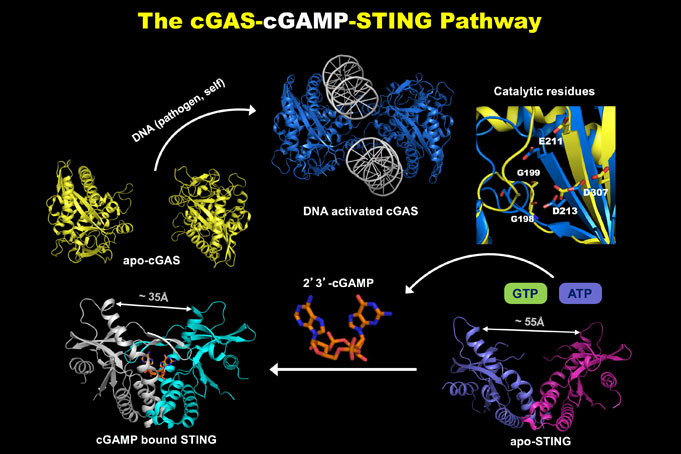
Project 1: Dr. Chen and his team are working at the molecular level exploring the cGAS-STING pathway and molecules that can activate that pathway in innate immunity, as well as studying the bridge to adaptive immunity.
He also is studying how to enhance the anti-tumor innate immune response with radiation and/or with other small molecules. Dr. Chen’s project includes searching for additional molecules with the potential to improve the innate immune response to the tumor.
Project 2: Dr. Fu is the primary investigator on the overall grant as well as leader of one of the three subprojects. His animal studies will focus on the cancer cell’s resistance to therapies on a more systemic level. The team is searching for ways to block the tumors’ resistance pathways to make combination cancer therapy more successful.
“I am looking more mechanistically at how radiation might be bridging the gap from DNA damage to activation of the cGAS-STING pathway,” says Dr. Fu, Professor of Pathology, Immunology, and Radiation Oncology, who holds the Mary Nell and Ralph B. Rogers Professorship in Immunology. “At the same time, I will study the cancer cells’ response. Many cancer therapies fail because of the cancer cell’s resistance – tumors figure out a way to resist the body’s immune response.”
Project 3: Dr. Hannan and his team are investigating changes in the tumor microenvironment – the area around the tumor – resulting from activation of the cGAS-STING pathway. His project will investigate the pathway’s relationship to neutrophil recruitment and the bridge from the innate to adaptive immune responses. He also will study how to use an adaptive immune response to induce a durable anti-tumor effect. This project will have access to post-treatment tissue from patients, which can be used to confirm preclinical findings.
In combination, the three projects have the potential to deepen the understanding of the interactions between the innate and adaptive immune systems, to significantly enhance cancer treatments, and, ultimately, to improve patient outcomes.
Dr. Chen, who is also a Professor in the Center for the Genetics of Host Defense, holds the George L. MacGregor Distinguished Chair in Biomedical Science.


